Mar. 9, 2018 Research Highlight Biology
Strengthening cancerous cell junctions with RhoA-stimulating drugs
Creating stronger seals between tissue surface cells could prevent cancers from spreading
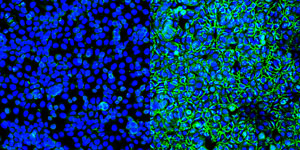 Figure 1: Human colon cells HT29 that have been treated (right) with the microtubule polymerization inhibitor nocodazole show many more green junction proteins holding the tissue together than those that were not treated (left). Bolstering these proteins could help stop cancerous cells from breaking off and travelling to other parts of the body to form secondary cancers. © 2018 RIKEN Center for Developmental Biology
Figure 1: Human colon cells HT29 that have been treated (right) with the microtubule polymerization inhibitor nocodazole show many more green junction proteins holding the tissue together than those that were not treated (left). Bolstering these proteins could help stop cancerous cells from breaking off and travelling to other parts of the body to form secondary cancers. © 2018 RIKEN Center for Developmental Biology
Metastatic cancer cells have a weak grip on each other, which creates the opportunity for them to spread to other parts of the body. By understanding how to strengthen the bonds between these cells, a team led by Masatoshi Takeichi at the RIKEN Center for Developmental Biology has revealed targets to help stop cancers from spreading1.
Many organ surfaces are composed of sheets of epithelial cells, knitted together by tightly sealed junctions made up of multi-protein complexes, including ‘apical-junctional complexes’. In cancerous tissue, these structures often fail to assemble properly, which can result in cancerous cells detaching, travelling and stimulating secondary cancers, a process known as metastatic growth.
Many cancerous cells still produce the correct junction building blocks but are unable to form normal junctions “even though they express key cell−cell adhesion molecules”, Takeichi says. “We suspected that junction formation is physiologically blocked and that their junctions could be restored by proper pharmacological treatments of the cells.”
His group at RIKEN tested this hypothesis by exposing human colon cancer cells with junction-forming defects to more than 150,000 different compounds. A tiny fraction of these proved capable of restoring normal epithelial cell organization—remarkably, many of them belonged to a particular class of compounds known as microtubule polymerization inhibitors, or MTIs. MTIs directly interfere with the formation of microtubules, protein-based cellular structures important to a number of processes. Masatoshi Takeichi and his team have found a pathway that could prevent metastatic growth in cancer by stimulating the formation of tight seals between epithelial cells © 2018 RIKEN
Masatoshi Takeichi and his team have found a pathway that could prevent metastatic growth in cancer by stimulating the formation of tight seals between epithelial cells © 2018 RIKEN
Takeichi and his colleagues then explored the interaction between microtubules, epithelial cells and MTI and discovered that the key to normal epithelial binding is a signaling cascade set in motion by MTI’s disassembly of microtubules. MTI treatment stimulated a sharp increase in levels of a protein called RhoA, which promotes apical-junctional complex formation by triggering extensive reorganization of other structural proteins, including myosin-IIA.
“It seems that colon carcinoma cells are defective in that they require more RhoA activity to organize epithelial-specific cell−cell junctions,” says Takeichi.
The takeaway is that it is hypothetically feasible to strengthen cancerous cell junctions with drugs that stimulate RhoA. However, mechanisms other than using MTIs would have to be found, given that microtubules perform many important functions in healthy cells and so inhibiting them could be highly toxic to the body.
Takeichi adds that there a still several steps before the RhoA mechanism could be tested in clinical trials. “We need to test whether artificially induced junction formation indeed affects invasion and metastasis of cancer cells in the body,” he says.
Related content
References
- 1. Ito, S., Okuda, S., Abe, M., Fujimoto, M., Onuki, T., Nishimura, T. & Takeichi, M. Induced cortical tension restores functional junctions in adhesion-defective carcinoma cells. Nature Communications 8, 1834 (2017). doi: 10.1038/s41467-017-01945-y
