Oct. 12, 2018 Research Highlight Biology
Gene Fam60a found to play a key role in the developing embryo
Healthy development of an embryo is found to depend on a protein that regulates the DNA of stem cells
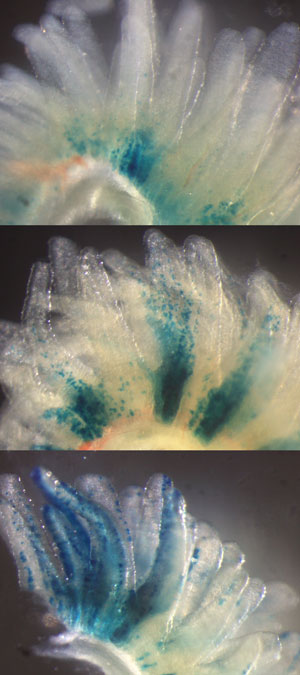 Figure 1: Over the course of several days, Fam60a-expressing stem cells (blue) proliferate and migrate to the outer edges of the finger-like villi that line the developing intestine in mouse embryos. Images show day 1 (top), 3 (middle) and 5 (bottom) after the onset of Fam60a expression. Reproduced from Ref. 1 and licensed under CC BY 4.0 © 2018 R. Nabeshima et al.
Figure 1: Over the course of several days, Fam60a-expressing stem cells (blue) proliferate and migrate to the outer edges of the finger-like villi that line the developing intestine in mouse embryos. Images show day 1 (top), 3 (middle) and 5 (bottom) after the onset of Fam60a expression. Reproduced from Ref. 1 and licensed under CC BY 4.0 © 2018 R. Nabeshima et al.
A gene with a central role in healthy embryonic development has been identified by RIKEN researchers1. It seems to control when and where various other genes are switched on or off, thereby controlling stem cell function in several critical organs.
This investigation of the gene, known as Fam60a, has its roots in a long-running effort to study pluripotency—the property that enables embryonic stem cells to give rise to many other cell types. “More than 20 years ago, we were interested in genes involved in pluripotency, and Fam60a was one of these,” says Hiroshi Hamada of the RIKEN Center for Biosystems Dynamics Research, who led the team with Shigehiro Kuraku, who is also at the center. “It’s highly expressed in embryonic stem cells, and its expression is down-regulated upon differentiation.” However, the specific nature of this gene’s function had remained enigmatic.
The Fam60a gene codes for an eponymous protein, so the researchers addressed this question by looking for molecular partners that the Fam60a protein interacts with in developing embryos. These experiments revealed that Fam60a is part of a larger multiprotein assembly known as Sin3a−Hdac, which is well known as a regulator of gene expression during development.
The team subsequently used a labeling strategy to monitor the expression of the Fam60a gene during embryogenesis. They saw high levels of Fam60a activity throughout the embryo around day nine of development, but expression subsequently became localized to tissues including the nervous system, lung and intestine (Fig. 1). “It was specifically expressed in actively dividing cells with stem cell characteristics,” says Hamada.
Critically, mice lacking this gene generally perished before birth, and the unviable embryos contained organs that were markedly underdeveloped and abnormally structured. Molecular analysis of these animals relative to mice with normal levels of Fam60a revealed that this protein contributes to the regulation of hundreds of genes. More specifically, Fam60a appears to bind to specific genomic regions as part of the Sin3a−Hdac complex, at which point it introduces chemical modifications to the DNA that directly influence whether adjacent genes are turned on or off.
Having established the importance of Fam60a to organ formation during embryonic development, Hamada now intends to explore whether this gene continues to play an important role later in life. “I’d like to examine whether Fam60a is expressed in somatic stem cells of other organs, such as neural stem cells found in the adult brain,” he says, “and what happens to these organs if Fam60a is lost in adulthood.”
Related contents
References
- 1. Nabeshima, R., Nishimura, O., Maeda, T., Shimizu, N., Ide, T., Yashiro, K., Sakai, Y., Meno, C., Kadota, M., Shiratori, H. et al. Loss of Fam60a, a Sin3a subunit, results in embryonic lethality and is associated with aberrant methylation at a subset of gene promoters. eLIFE 7, e36435 (2018). doi: 10.7554/eLife.36435
