Nov. 22, 2007 Research Highlight Chemistry
Hidden surface chemistry revealed
A new technique that works at normal pressures shows molecular interactions at liquid interfaces
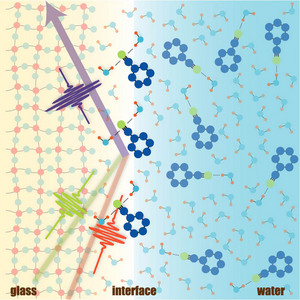 Figure 1: Molecules with a CN group at a fused silica glass/water interface. pi-type H-bonds that are not found in bulk liquid phases, where sigma-type H-bonds prevail, were detected at the glass/ water interface using χ(4) Raman spectroscopy.
Figure 1: Molecules with a CN group at a fused silica glass/water interface. pi-type H-bonds that are not found in bulk liquid phases, where sigma-type H-bonds prevail, were detected at the glass/ water interface using χ(4) Raman spectroscopy.
RIKEN scientists have developed a technique that opens a window into a previously shadowy realm—the point where liquids and solids meet.
“Liquid interfaces play crucial roles in many phenomena, but only little is understood about what really happens at the interface,” explains Tahei Tahara of RIKEN’s Discovery Research Institute in Wako. Whether studying how a solid catalyst speeds up an industrially useful chemical reaction, or watching the biochemistry inside a human cell, understanding liquid–liquid and solid–liquid interfaces is crucial.
But studying the chemistry of submerged surfaces at a molecular scale is not trivial. “Most of the surface analysis techniques need a high vacuum,” says Tahara. “They cannot be applied to liquid interfaces, because liquids cannot be kept in the vacuum.”
Tahara and his colleague, Shoichi Yamaguchi, have now developed an alternative technique that works at normal pressures, based on a form of spectroscopy called χ(4) Raman1. This uses visible and near-infrared light to vibrate molecules at interfaces, and then detects the light they emit as they return to their normal state. The emitted light carries information about the strength of the chemical bonds that hold the molecule together, and also how these molecules interact with their neighbors at the interfaces.
In Tahara and Yamaguchi’s method, ultrashort laser pulses of light interact with a sample material four times in quick succession. This allows them to selectively observe only those molecules which lie at interfaces, says Tahara.
To test their method, the scientists dissolved a dye molecule called rhodamine 800 in water and mixed in a pure form of sand called silica glass.
They used χ(4) Raman to measure the stretchiness of the chemical bond that connects a particular carbon atom to a nitrogen atom (forming a CN group) within rhodamine. This in turn revealed the pattern of water molecules that were assembled around it (Fig. 1).
In water, rhodamine normally has many water molecules forming a network around the nitrogen atom of the CN group. But by comparing their experimental results with theoretical calculations, they found that this pattern changed as rhodamine approached a solid fragment of silica, until just one or two water molecules were connected to the bond between nitrogen and carbon.
“Our finding is the first step that clearly shows that the fashion of molecular interaction is significantly different at the interface,” says Tahara. Being able to study this difference should help scientists interested in areas as diverse as catalysis and biology.
References
- 1. Yamaguchi, S. & T. Tahara, T. χ(4) Raman spectroscopy for buried water interfaces. Angewandte Chemie International Edition 46, 7609–7612 (2007). doi: 10.1002/anie.200701609
