Dec. 26, 2016 Press Release Biology Chemistry
Using super-slow motion movie, scientists pin down the workings of a key proton pump
Using powerful new tools, scientists from the RIKEN SPring-8 Center and collaborators have demonstrated how bacteriorhodopsin, a "proton pump", uses light to transport protons across the cell membrane to create a charge difference that can be used to power a cell's activities.
Bacteriorhodopsin is a protein that absorbs light and transports protons across cell membranes—a key function of biological systems. But scientists have long wondered how it does this, and how it can push protons in a single direction, from the inside to the outside of the cell.
To find out, the scientists turned to SACLA, a powerful x-ray laser that produces rays a million times brighter than conventional synchrotron facilities. These x-rays can be used to determine the structure of proteins and other molecules passed through the beam. Because the beam is so strong, however, it vaporizes the sample instantly so it is important to get images before it is destroyed, a trick known as "diffraction before destruction."
For the current study, the team used a technique known as time-resolved serial femtosecond crystallography, where they took thousands of images of bacteriorhodopsin at various time points after it was struck by light, based on a "pump-probe method." Putting this all together, they were able to piece together the story of how the membrane protein is able to move protons against a gradient into the more positively charged environment outside the cell, creating a charge like a battery that could be used to power chemical reactions.
Says Eriko Nango, the first author of the study, published in Science, "With this work, we were able to shed light on the mechanism of proton transfer and bring closure to a long-standing debate regarding the mechanism. The photoexcitation leads to a change of conformation in retinal, the key part of bacteriorhodopsin that absorbs the light. This in turns leads to the displacement of amino acid residues above the retinal towards the cytoplasm, and a transient water appears in that space to assist the primary proton transfer. Following this, a delicate molecular cascade prevents the proton from moving backward, and it is pushed up outside of the cell.”
Studies using previous methods had identified many of the steps, but because of suspicions that the radiation itself might be causing changes, it was impossible to conclusively show the precise mechanism. With the new technique, the group finally pinned this down.
According to So Iwata, who led the team, "new techniques using powerful x-ray lasers are giving us insights into precisely how processes such as proton pumping take place in actual biological systems. This will give us much greater understanding of these actions, and ultimately lead to insights that will allow us to manipulate them more effectively."
The work was done by RIKEN scientists in collaboration with researchers from the University of Gothenburg, Institut de Biologie Structurale, Paul Scherrer Institute, Kyoto University, the University of Tokyo and other institutes.
Reference
- Eriko Nango et al., A three dimensional movie of structural changes in bacteriorhodopsin, Science, doi: 10.1126/science.aah3497
Contact
Group Director
So Iwata
SACLA Science Research Group
Photon Science Research Division
RIKEN SPring-8 Center
Jens Wilkinson
RIKEN International Affairs Division
Tel: +81-(0)48-462-1225 / Fax: +81-(0)48-463-3687
Email: pr@riken.jp
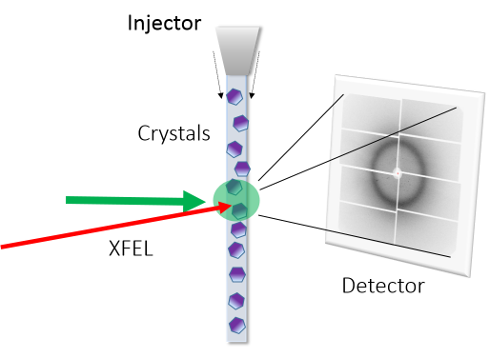
Setup of the experiment
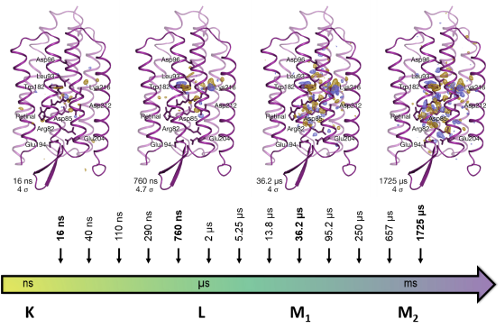
Changes in bacteriorhodopsin over time
