Jan. 11, 2017 Press Release Biology Medicine / Disease
New transplant technique restores vision in mice
Researchers at the RIKEN Center for Developmental Biology in Kobe, Japan have shown that retinal transplants derived from induced pluripotent stem cells (iPSCs) can restore visual function in mice. Published in Stem Cell Reports, the study shows that neurons in the implanted retinal sheets integrate well with the host, respond to light, and send their signals to the brain as judged by behavioral experiments.
Retinal degeneration is mostly a hereditary disease that is characterized by the death of photoreceptors—the light-sensitive neurons in the eye—which eventually leads to blindness. While many have attempted to treat the disease through retinal transplants, and some have shown that transplanting graft photoreceptors to the host without substantial integration can rescue retinal function, until now, no one has conclusively succeeded in transplanting photoreceptors that functionally connect to host cells and send visual signals to the host retina and brain.
The team led by Masayo Takahashi studied this problem using a mouse model for end-stage retinal degeneration in which the outer nuclear layer of the retina is completely missing. This is an important issue because in clinical practice this type of therapy would most likely target end-stage retinas in which of the photoreceptors are dead and the next neurons up the chain do not have any input.
The Takahashi group has recently shown that 3D retinal sheets derived from mouse embryonic stem cells develop normal structure connectivity. “Using this method was a key point,” explains first author Michiko Mandai, “Transplanting retinal tissue instead of simply using photoreceptor cells allowed the development of more mature, organized morphology, which likely led to better responses to light.”
In order to assess the success of the transplantations, the team integrated some modifications to the retinal sheets and the model mice. They used a fluorescent protein to label the ends of the photoreceptors, which is where they would connect to the host neurons—the bipolar retinal cells—and ultimately the brain. After labeling the host retinal bipolar cells with a different fluorescent protein, they found that the labeled cell terminals from the graft did indeed make contact with the cells labeled in the host, indicating that the newly grown photoreceptors naturally connected themselves to the bipolar cells in the model mice.
To assess whether the mice could see light, the researchers used a behavioral learning task. Mice with normal vision can learn to associate sounds or light with different events, the same way that Pavlov’s famous dog associated food with the sound of a bell. While the model mice who lacked a photoreceptor layer in their retinas could not learn to associate anything with light before surgery, they could after the transplant, provided that a substantial amount of the transplant was located in the correct place. This means that not only did the new cells in the retina respond to light, but the information traveled to the brain and could be used normally to learn.
“These results are a proof of concept for using iPSC-derived retinal tissue to treat retinal degeneration,” says Mandai. “We are planning to proceed to clinical trials in humans after a few more necessary studies using human iPSC-derived retinal tissue in animals. Clinical trials are the only way to determine how many new connections are needed for a person to be able to ‘see’ again.”
Although the results are promising, Mandai cautions that for now this therapy is at the developmental stage. “We cannot expect to restore practical vision at the moment,” She explains. “We will start from seeing a simple light, then possibly move on to larger figures in the next stage.”
Reference
- Michiko Mandai, Momo Fujii, Tomoyo Hashiguchi, Genshiro A Sunagawa, Shinichiro Ito, Jianan Sun, Jun Kaneko, Junki Sho, Chikako Yamada, Masayo Takahashi, "iPSC-derived retinal transplants improve vision in rd1 end-stage retinal degeneration mice", Stem Cell Reports
Contact
Project Leader
Masayo Takahashi
Laboratory for Retinal Regeneration
RIKEN Center for Developmental Biology
Adam Phillips
RIKEN International Affairs Division
Tel: +81-(0)48-462-1225 / Fax: +81-(0)48-463-3687
Email: pr@riken.jp
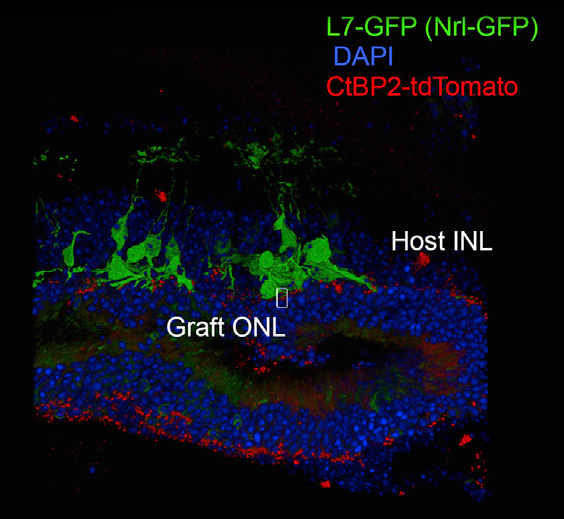
Synaptic integration of graft retina into model mouse
