Aug. 2, 2019 Research Highlight Biology
Pre- and postsynaptic sites regulate synaptic strength differently
The differential regulation of neuronal signal strength in pre- and postsynaptic cells contributes to information processing in the brain
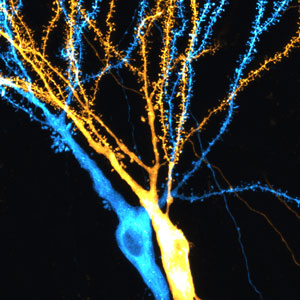 Figure 1: Dendrites of hippocampal neurons receive and process information from other neurons in the brain. © 2019 RIKEN Center for Brain Science
Figure 1: Dendrites of hippocampal neurons receive and process information from other neurons in the brain. © 2019 RIKEN Center for Brain Science
The regulation of synaptic strength in response to activity differs for pre- and postsynaptic sites in mice, RIKEN neuroscientists have found1. This finding provides evidence for a neural mechanism that compensates for presynaptic plasticity.
Neurons receive, integrate and transmit information as electrical or chemical signals. Importantly, neurons can alter the strength of their connections, or synapses, in response to the inputs they receive. This synaptic plasticity controls how effectively two neurons communicate with each other and is crucial for memory formation, consolidation and retrieval. However, little is known about how changes in individual synaptic strength are regulated within neurons and across networks.
Now, by combining electrophysiological recordings with live-cell imaging of pyramidal neurons in the hippocampus of mice, Yukiko Goda at the RIKEN Center for Brain Science, Japan, and Mathieu Letellier at the University of Bordeaux, France, have assessed the strengths of individual synapses and explored how they are modified by neural activity in relation to neighboring synapses.
They found that the efficacy of neurotransmitter release from presynaptic sites, which correlates with presynaptic strength, depends on both the identity of the presynaptic cell and the location of the presynaptic site along the dendrite. A branch of the tree-like extension in the postsynaptic cell, the dendrite receives the input from other cells (Fig. 1). By contrast, the postsynaptic strengths of individual synapses, determined from dendritic spine size, depended less on the identity of the presynaptic cell and mainly on local synaptic contacts along the dendritic branch of the postsynaptic cell.
These findings highlight a role for local interactions between postsynaptic compartments along the dendritic branch that could lead to the sharing of plasticity-related molecules and spine clustering or, alternatively, to competition for such molecules and an overall reduction in postsynaptic strengths. The resulting synaptic strength distribution in the dendrite will ultimately determine how each input is integrated in the postsynaptic neuron to produce an output signal.
The researchers also showed that presynaptic cell stimulation, which led to a normalization of presynaptic strengths, whereby synapses with a low initial neurotransmitter release probability showed a large increase in this parameter and vice versa, was accompanied by a decrease in postsynaptic strengths.
This mismatch between the regulation of synaptic strength at pre- and postsynaptic sites was unexpected. “Our findings indicate that the directions of synaptic strength changes between the pre- and postsynaptic sites are not necessarily parallel and may represent a means to preserve network stability without compromising the specificity of incoming information,” Goda explains.
The team is now seeking to determine how activity-dependent synaptic strength regulation produces effective signal integration and processing. “We aim to understand the relevance of synaptic strength tuning to hippocampal memory circuits and make this knowledge available for the design of efficient artificial networks,” she adds.
Related contents
- Making the right connections
- Unearthing the pathways of plasticity
- Change in the brain: astrocytes finally getting the recognition they deserve
References
- 1. Letellier, M., Levet, F., Thoumine, O. & Goda, Y. Differential role of pre- and postsynaptic neurons in the activity-dependent control of synaptic strengths across dendrites. PLoS Biology 17, e2006223 (2019). doi: 10.1371/journal.pbio.2006223
