Feb. 23, 2007 Research Highlight Biology
Steering mechanism for nerve circuit formation revealed
A recent study extends our understanding of how nerve cells reach out towards attractive stimuli
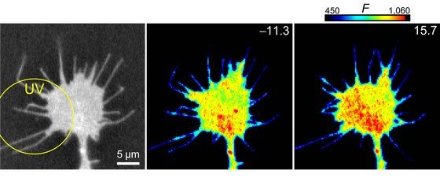 Figure 1: Vesicle fusion is enhanced preferentially in areas of calcium flux. © Nature Neuroscience/ Nature Publishing Group/ 10/ 62 (2006).
Figure 1: Vesicle fusion is enhanced preferentially in areas of calcium flux. © Nature Neuroscience/ Nature Publishing Group/ 10/ 62 (2006).
New work sheds light on the processes responsible for guiding the tips of nerve cells in the right direction. Nerve cells receive messages called impulses through one end, the dendritic tree, and dispatch impulses out the other end, the axon. To ensure that messages reach the desired destination, nerve cells send out axons that elongate toward their appropriate targets. Growth cones, the tips of these axons, ‘sense’ the location of the targets. In turn, targets make their presence known by sending appropriate positional cues.
Previous work indicates that these cues trigger fluxes of calcium within growth cones. A team led by Hiroyuki Kamiguchi, a scientist at RIKEN’s Brain Science Institute in Wako, identified events linking calcium flux with directional movement of growth cones1.
The researchers hypothesized that the direction of axon elongation might shift with preferential addition of new membrane material to one side of the growth cone. Testing this hypothesis required tracking the movement of vesicles, which are sources of new membrane material found within nerve cells, after calcium levels increase in specific regions within the growth cone.
Indeed, nerve cell vesicles labelled with a fluorescent molecule preferentially migrated towards calcium released on one side of the growth cone. This directed-trafficking of vesicles required microtubules, which transport cargo within many cell types.
Vesicles moving in the direction of calcium release contained VAMP2, a member of the SNARE family of proteins, which act like ‘zippers’ to enhance fusion between two closely opposed membranes. The researchers observed such fusion of the migrated vesicles with surface membranes of the growth cone (Fig. 1). Application of tetanus neurotoxin, which prevents VAMP2-directed fusion, prevented the growth cone turning in the direction of calcium release.
Importantly, VAMP2 inhibition also blocked the growth cone turning towards myelin-associated glycoprotein and nerve growth factor, which are calcium-dependent ‘natural’ stimuli encountered by growth cones within the body.
Identification of the steps linking calcium flux to vesicle movement remains for future study. Regardless, these data provide another link in the chain of events responsible for dispatching nerve cell messages in the correct direction.
“This work provides a simple answer to the long-lasting question of how a growth cone changes its direction of locomotion. Further investigation of steering mechanisms will enable us to manipulate the direction of axon elongation within the body. This technology could contribute to the development of therapeutic strategies by which regenerating axons can be guided toward appropriate targets to achieve functional recovery after nervous system injury,” says Kamiguchi.
References
- 1. Tojima, T., Akiyama, H., Itofusa, R., Li, Y., Katayama, H., Miyawaki, A. & Kamiguchi, H. Attractive axon guidance involves asymmetric membrane transport and exocytosis in the growth cone. Nature Neuroscience 10, 58–66 (2006). doi: 10.1038/nn1814
