Aug. 1, 2008 Research Highlight Biology
Tracing lymphocyte development signaling pathways
Two related proteins are essential for proliferation and survival of immune cell precursors
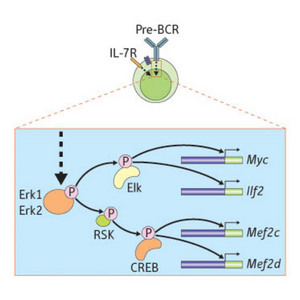 Figure 1: The proteins Erk1 and Erk2 control a transcription factor network that allows B lymphocyte progenitors to traverse the pre-BCR developmental checkpoint.
Figure 1: The proteins Erk1 and Erk2 control a transcription factor network that allows B lymphocyte progenitors to traverse the pre-BCR developmental checkpoint.
Researchers have pinpointed the proteins required for transduction of signals directing the development of B lymphocytes, a type of immune cell. Responsible for producing antibodies capable of neutralizing invading microbes, B lymphocytes develop in the bone marrow and, in mature form, circulate throughout the body through the blood and lymphoid organs.
Before exiting the bone marrow, B lymphocyte precursors are propelled through a series of distinct developmental stages. Progression through early stages depends largely on signals triggered by extracellular growth factors, whereas passage through some later stages requires a signal from the pre-BCR, a receptor expressed on the surface of B lymphocyte precursors.
Pre-BCR signals culminate in the induction of gene transcription in the nucleus. Although the cell-surface proteins responsible for initiating pre-BCR signals have been identified, Tomoharu Yasuda and co-workers at RIKEN’s Research Center for Allergy and Immunology in Yokohama set out to identify proteins further down the pre-BCR signaling pathway.
The team focused on the related proteins Erk1 and Erk2, which transduce signals by phosphorylating, or ‘tagging’ downstream target proteins. Using genetic techniques, the researchers generated mice in which Erk1, Erk2 or both Erk1 and Erk2 are deleted or ‘knocked out’ on demand1.
Compared to normal mice or those lacking either Erk1 or Erk2, mice lacking both Erk1 and Erk2 exhibited an almost complete block in development beyond the stage during which pre-BCR signaling occurs. Despite normal surface pre-BCR expression, these ‘double knockout’ precursors showed impaired proliferation and survival.
Consistent with the defects of the double-knockout cells, B lymphocyte precursors treated with an inhibitor of Erk activity expressed lower amounts of immediate early genes (IEG), which are required for population expansion.
As Erk1 and Erk2 do not directly bind to DNA, the team searched for DNA-binding factors that link Erk proteins with IEG upregulation. Noting the presence of binding sites for the factors Elk and CREB in the DNA regions controlling IEG expression, the team showed that pre-BCR-induced Erk activation results in Elk and CREB phosphorylation (Fig. 1).
Illustrating the importance of Erk activity in B lymphocyte development, forced expression of mutant forms of Elk and CREB that lack Erk phosphorylation sites blocked pre-BCR-induced population expansion.
“Pre-BCR signals transmitted through Erk and downstream transcription factors are critical for pre-B cell proliferation,” says Yasuda. “Drugs able to promote or suppress such molecular activity may be useful for treatment of acute lymphobastic leukemia or immune deficiency disorders.”
References
- 1. Yasuda, T., Sanjo, H., Pagès, G., Kawano, Y., Karasuyama, H., Pouysségur, J., Ogata, M. & Kurosaki, T. Erk kinases link pre-B cell receptor signaling to transcriptional events required for early B cell expansion. Immunity 28, 499–508 (2008). doi: 10.1016/j.immuni.2008.02.015
