Jan. 29, 2010 Research Highlight Biology
A window to embryonic development
Cell-cycle progression in fish embryos can now be tracked visually in three dimensions
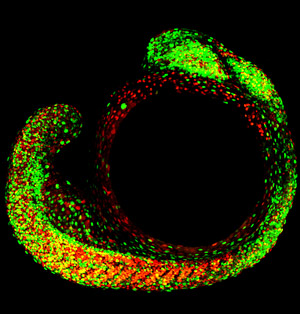 Figure 1: A view of cell proliferation versus differentiation, using Fucchi technology, in a whole zebrafish embryo during segmentation at 18 hours post-fertilization. The retina, full of green nuclei, is visible at the top of the image and the notochord is covered by skeletal muscles along the bottom. © (2009) RIKEN
Figure 1: A view of cell proliferation versus differentiation, using Fucchi technology, in a whole zebrafish embryo during segmentation at 18 hours post-fertilization. The retina, full of green nuclei, is visible at the top of the image and the notochord is covered by skeletal muscles along the bottom. © (2009) RIKEN
A RIKEN-led team of molecular biologists has developed a visual means of tracking DNA replication as well as cell division and movement in transparent, whole zebrafish embryos. The technique could eventually lead to three-dimensional reconstructions of early development at a cellular level. The researchers already have used their technology to study the cellular development of the retina and of the primitive backbone or notochord, in which they found cell-cycle progression occurring in waves from head to tail.
The team, which is based at the RIKEN Brain Science Institute in Wako but includes researchers from other Japanese institutes and universities, had previously developed technology for tracing the cell cycle in mammalian cells. They called it Fucci—fluorescent ubiquitination-based cell cycle indicator.
Fucci technology utilizes reciprocal fluctuations in the levels of two proteins involved in regulating the cell cycle, Cdt1 and geminin. Cdt1 levels are highest just before DNA replication, whereas geminin levels build up during DNA synthesis and fall during the phase between divisions. The destruction of each of these proteins is preceded by tagging with the protein ubiquitin.
With the Fucci technology, the researchers attach a fluorescent marker to the part of the proteins to which ubiquitin links—orange for Cdt1 and green for geminin. Thus the cycle can be followed by visually tracking the fluctuation of orange and green in the cell. In the opaque mouse embryo, however, this can only be done in slices, not in the whole embryo in three dimensions. So they decided to transfer their technology to zebrafish whose early embryos are transparent. They found, however, that the original Cdt1Fucci tag did not work because of differences in the ubiquitin-mediated degradation system between mouse and zebrafish embryos.
To overcome this problem, the researchers designed and constructed a Cdt1 marker for the fish cells, and using that experience, also improved the geminin marker. After testing in cultured cells to determine that the tags worked and did not affect cell performance, they then created a zebrafish line, dubbed Cecyil, with the markers incorporated genetically.
Using their Fucci zebrafish, the researchers were able to observe development of the retina and differentiation of the notochord (Fig. 1). Their results are published in a recent paper in the Proceedings of the National Academy of Sciences1.
“Remarkably, we discovered two waves of cell-cycle transitions traveling from anterior to posterior of the notochord,” says project leader Atsushi Miyawaki. “In future we hope to develop indicators for different cell-cycle phases and other animals.”
References
- 1. Sugiyama, M., Sakaue-Sawano, A., Iimura, T., Fukami, K., Kitaguchi, T., Kawakami, K., Okamoto, H., Higashijima, S. & Miyawaki, A. Illuminating cell-cycle progression in the developing zebrafish embryo. Proceedings of the National Academy of Sciences USA Published online before print, 18 November 2009. doi: 10.1073/pnas.0906464106
