Jul. 13, 2012 Research Highlight Biology
Making live cell microscopy affordable
An inexpensive and non-destructive microscopy method for illuminating living cells brings advanced biology within reach of under-resourced laboratories
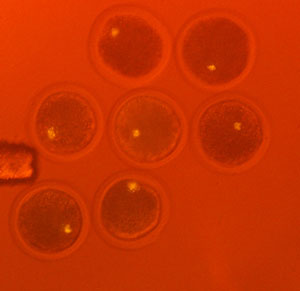 Figure 1: An image of fluorescently labeled unfertilized cow oocytes using the halogen lamp filter adapter. The nuclei are clearly observed at the center of the cells. Reproduced, in part, from Ref. 1 © 2012 K. Yamagata et al., RIKEN Center for Developmental Biology
Figure 1: An image of fluorescently labeled unfertilized cow oocytes using the halogen lamp filter adapter. The nuclei are clearly observed at the center of the cells. Reproduced, in part, from Ref. 1 © 2012 K. Yamagata et al., RIKEN Center for Developmental Biology
Researchers in Japan have developed a low-intensity light source that allows cell biologists to visualize and handle live cells without destroying them during prolonged exposure1. In addition to laying the foundation for new cell manipulations, the development will make advanced biology requiring fluorescence microscopy accessible to underfunded laboratories. Led by Teruhiko Wakayama from the RIKEN Center for Developmental Biology, Kobe, the researchers developed an adapter, equipped with a halogen lamp, for a conventional microscope.
Typically, conventional microscopes used in live cell studies rely on powerful ultraviolet (UV) lamps or lasers to illuminate cells labeled with a fluorescent dye molecule. However, extended imaging times or continuous exposure to these high-intensity light sources results in harmful effects to cells. Although the problem seemed complex, Wakayama explains that simply lowering the strength of the light source via a halogen lamp solved the problem of phototoxicity.
The adapter developed by the researchers consisted of a small excitation filter and a diaphragm that allowed some light to leak around its periphery. They placed it on top of the microscope’s condenser lens, which concentrates light from the lamp onto the samples. By closing the diaphragm, they channeled all the light through the filter, yielding only fluorescence. Opening the diaphragm produced a bright field image that merged with the fluorescent signal. The researchers found that they could tune the relative intensities of both images by varying this opening.
Wakayama and his colleagues tested the performance of their device for monitoring the enucleation, or the removal of metaphase chromosomes, from female reproductive cells called oocytes. They discovered that, unlike traditional enucleation approaches, the new method allowed them to successfully remove the chromosomes in quantitative yields. This made it unnecessary to use additional analytical techniques to confirm the absence of chromosomes (Fig. 1). Overall, compared to conventional approaches, the halogen lamp method significantly simplified fluorescent observation of the enucleation procedure and reduced processing times without affecting cell viability.
The team is currently planning to use their fluorescence imaging system to rapidly detect the fertilization of human oocytes. “Previously, we could not observe these oocytes using fluorescence microscopy [owing] to UV lamp-induced cell damage, but now we can thanks to our adapter,” says Wakayama. Moreover, the researchers are proposing to introduce their device to high schools, which use simple microscopes that lack fluorescence imaging capabilities. “Our system will make it possible for all students to have access to advanced fluorescence microscopy without excessive costs,” he adds.
References
- 1. Yamagata, K., Iwamoto, D., Terashita, Y., Li, C., Wakayama, S., Hayashi-Takanaka, Y., Kimura, H., Saeki, K. & Wakayama, T. Fluorescence cell imaging and manipulation using conventional halogen lamp microscopy. PLoS ONE 7, e31638 (2012). doi: 10.1371/journal.pone.0031638
