Jun. 1, 2012 Research Highlight Biology
Old cells learn new tricks
As certain neurons within the hippocampus of the brain mature, their contributions to memory and perception change
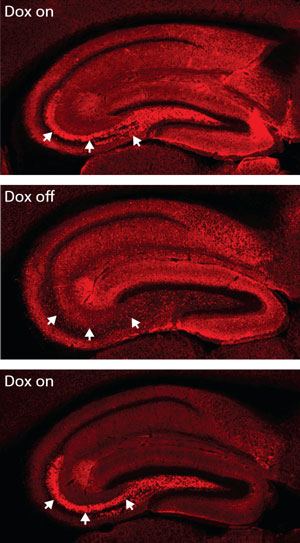 Figure 1: Fluorescently labeled VAMP2 protein (red) offers a useful indicator of synaptic activity. Granule cells in the dentate gyrus of the transgenic mice can successfully transmit signals to the CA3 region of the brain when treated with the chemical activator doxycycline (Dox) (top; arrows). Removal of Dox (middle) can inactivate this signaling, and be reactivated through the re-application of Dox (bottom). Reproduced, with permission, from Ref. 1 © 2012 Elsevier
Figure 1: Fluorescently labeled VAMP2 protein (red) offers a useful indicator of synaptic activity. Granule cells in the dentate gyrus of the transgenic mice can successfully transmit signals to the CA3 region of the brain when treated with the chemical activator doxycycline (Dox) (top; arrows). Removal of Dox (middle) can inactivate this signaling, and be reactivated through the re-application of Dox (bottom). Reproduced, with permission, from Ref. 1 © 2012 Elsevier
The philosopher George Santayana famously remarked that “Those who cannot remember the past are condemned to repeat it.” The hippocampus is essential to avoiding such mistakes, as this brain structure helps process memories in a manner that can usefully guide an organism’s behavior.
The brain uses its ability to recall past actions to identify differences between highly similar yet distinct events, such as when you leave your keys in a different place than usual—a process called ‘pattern separation’. The hippocampus also performs the opposite function—known as ‘pattern completion’—in which it uses isolated pieces of information to reconstruct stored memories; looking at an old photo, for example, will cause the hippocampus to summon thoughts from the day the photo was taken.
Neuroscientists have generally believed that distinct segments of the hippocampus are responsible for pattern separation and completion. “These two functions have been thought to be opposing and competing processes,” explains Toshiaki Nakashiba, a researcher with Susumu Tonegawa’s team at the RIKEN-MIT Center for Neural Circuit Genetics in Cambridge, Massachusetts, USA. “Previous studies have indicated that the dentate gyrus is the anatomical location in the hippocampus for pattern separation, while the CA3 region handles pattern completion.”
Tonegawa, Nakashiba and colleagues, however, have demonstrated that both processes are managed within the dentate gyrus, with specific contributions from two distinct subsets of neurons known as granule cells (GCs)1.
Age discrimination
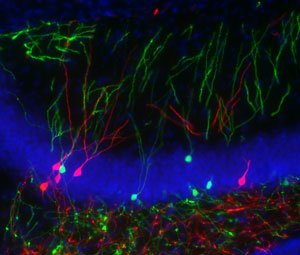 Figure 2: Granule cells within the dentate gyrus play distinct roles in memory formation and recall as they age (green, old granule cells; red, young granule cells). © 2012 Toshiaki Nakashiba, RIKEN-MIT Center for Neural Circuit Genetics
Figure 2: Granule cells within the dentate gyrus play distinct roles in memory formation and recall as they age (green, old granule cells; red, young granule cells). © 2012 Toshiaki Nakashiba, RIKEN-MIT Center for Neural Circuit Genetics
In some ways, the study benefited from a happy accident. The dentate gyrus is one of the only parts of the brain that replenishes neurons in adult life. At first, newly generated ‘young GCs’ are highly active, but gradually they settle down and become virtually indistinguishable from GCs formed before birth. The researchers developed genetically modified mice in which they could broadly inactivate, and re-activate, hippocampal GCs with a chemical agent that causes their synapses to stop working without harming the cells (Fig. 1). However, they were in for a surprise. “Unexpectedly, this mutant strain retained intact transmission from young GCs,” says Nakashiba. “We didn’t expect to see the selective blocking of synaptic transmission in ‘old’ granule cells, but this strain turned out to be a good model for studying the distinct roles of these cells in the dentate gyrus.”
Indeed, the subsequent observation of some unusual behavioral patterns in these mice led Nakashiba and his colleagues to design a series of tests that allowed them to characterize the relative contributions of these different generations of adult-born GCs to memory function.
In one set of experiments, the researchers tested pattern separation by training mice to associate a particular environment with a mildly painful shock. After several training blocks, wild-type mice learned to distinguish a shock chamber from a slightly different-looking chamber with no shock associated. Remarkably, mice with old GCs switched off not only achieved this discrimination, but did so faster, thus suggesting a primary role for young GCs in pattern separation.
Subsequent pattern separation experiments with more obviously different environments provided further support for this model. Mice with inactivated old GCs performed moderately worse at discrimination than wild-type counterparts; however, when the researchers used targeted irradiation to specifically kill off hippocampal young GCs, their performance suffered considerably.
Tonegawa’s team also found compelling evidence that older GCs play a key role in pattern completion. In one test, they placed mice in a maze filled with opaque water, with four objects placed at different positions to provide orientation cues. Using those cues, wild-type mice and transgenic animals quickly learned to find an invisible platform submerged below water level at a fixed position in the maze.
Subsequent inactivation of old GCs did not notably impair the ability of transgenic mice to locate the platform, as long as all four cues were present. However, when the researchers reduced the number of cues to one, the treated animals took markedly longer than wild-type mice to find the correct spot. This, together with other experiments, suggests that the selective inactivation of old GCs considerably reduces the speed of pattern completion in the hippocampus.
Balance of power
This latter result is particularly surprising because neuroscientists have generally believed that pattern completion is performed in CA3, a region of the hippocampus that receives information from the dentate gyrus and which is characterized by extensive interconnection between its neurons. “This was believed to be a good network for associating multiple information sources, such as spatial location and contextual information, into a coherent representation,” explains Nakashiba.
Instead, this process seems to be managed alongside pattern separation within the dentate gyrus (Fig. 2). The researchers raise the possibility that newly formed young GCs may be best-equipped to achieve the recognition of novel elements required for pattern separation, whereas older, more thoroughly integrated GCs are more likely to act as established sources of information for CA3 that facilitate memory recall through pattern completion.
Changes in cellular demographics could therefore have serious consequences. “Our data demonstrated that mice devoid of old neurons had enhanced pattern separation in some conditions, but were also defective in pattern completion,” says Nakashiba. “This suggests that the balance between pattern separation and completion could be altered as a result of the loss of old neurons.”
Tonegawa’s group is now attempting to zoom in on the GC subsets that contribute most to these processes, and Nakashiba anticipates that the results of these studies could ultimately have important implications for understanding the neurological roots of cognitive deficits associated with disease, brain damage or old age.
References
- 1. Nakashiba, T., Cushman, J.D., Pelkey, K.A., Renaudineau, S., Buhl, D.L., McHugh, T.J., Barrera, V.R., Chittajallu, R., Iwamoto, K.S., McBain, C.J.et al. Young dentate granule cells mediate pattern separation, whereas old granule cells facilitate pattern completion. Cell 149, 188–201 (2012). doi: 10.1016/j.cell.2012.01.046
About the Researcher
Toshiaki Nakashiba

Toshiaki Nakashiba was born in Kanagawa, Japan, in 1971. He graduated from the Faculty of Sciences, Kyoto University in 1995, and in 2001 obtained his PhD from the same university in the field of developmental neurobiology, supervised by Dr Shigeyoshi Itohara. Nakashiba then joined the laboratory of Dr Susumu Tonegawa at the Center for Learning and Memory at the Massachusetts Institute of Technology (MIT) in Cambridge, USA, and in 2004 was promoted to the position of research scientist at the RIKEN-MIT Center for Neural Circuit Genetics. His research focuses on the role of hippocampal circuits in learning and memory processes, which he observes through behavioral experiments conducted on genetically modified mice.
