Mar. 18, 2016 Research Highlight Chemistry
Breaking bonds for probes and drugs
A method for transforming carbon–fluorine bonds offers new opportunities in drug research and medical imaging
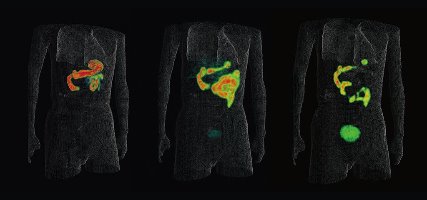 Figure 1: Positron emission tomography (PET) probes can be used to pinpoint the location of tumors in the body. RIKEN researchers have found a straightforward way to produce PET probes containing radioactive fluorine. © 2016 RIKEN Center for Life Science Technologies and Osaka City University
Figure 1: Positron emission tomography (PET) probes can be used to pinpoint the location of tumors in the body. RIKEN researchers have found a straightforward way to produce PET probes containing radioactive fluorine. © 2016 RIKEN Center for Life Science Technologies and Osaka City University
A chemical procedure developed by an all-RIKEN research team has the potential to enhance the usefulness of positron emission tomography (PET) for discovering new drugs and diagnosing diseases.
Many promising candidate drugs have to be abandoned when they enter clinical trials because they give rise to adverse side effects. Such side effects could be detected earlier by using improved methods to assess the metabolism of a drug candidate after administration. Incorporating suitable radioactive atoms―radioisotopes―into prospective drugs allows researchers to follow how a molecule is processed in the body. For example, PET scanning detects gamma-rays generated by the release of positrons when radioisotopes spontaneously decay.
“As the number of fluorine-containing drugs is increasing,” says Takamitsu Hosoya of the RIKEN Center for Life Science Technologies, “it is vital to find ways to incorporate radioactive fluorine atoms into a wide range of molecules.”
Compounds known as fluoroarenes are suitable starting materials for making fluorine-containing probes. But to transform them into useful probes requires breaking one of the strongest bonds in nature―the carbon–fluorine bond.
This is the key process that Hosoya and his colleagues have now achieved. They used a nickel–copper catalyst to break the carbon–fluorine bond in a way that permits non-radioactive fluorine-19 atoms to be swapped with radioactive counterparts, fluorine-18 atoms1.
This also permitted the researchers to transform the initial fluoroarenes into a range of other potentially useful compounds, including some that do not contain fluorine (Fig. 1).
The reaction schemes involve creating an organoboron compound, in which the original fluorine atom is replaced with a boron atom, prior to further chemical transformation. Organoboron compounds are widely used intermediates in chemical synthesis. Appreciating the details requires a deep knowledge of chemistry, but the key consequence is that fluoroarenes have now become a much more versatile source of many kinds of useful molecules.
“We anticipate that our method will be applied to make new PET probes that can help the early diagnosis of diseases,” Hosoya explains. Such PET probes are chemicals that are selectively taken up by diseased tissues, such as tumors, and thus reveal the location of such tissue in PET images.
The researchers plan to build on this work by linking it to existing collaborations with clinical teams that can use it in applied research in humans. “By developing our innovative chemical methods, we hope to contribute to the advance of life science and drug discovery,” says Hosoya.
References
- 1. Niwa, T., Ochiai, H., Watanabe, Y. & Hosoya, T. Ni/Cu-catalyzed defluoroborylation of fluoroarenes for diverse C–F bond functionalizations. Journal of the American Chemical Society 137, 14313–14318 (2015). doi: 10.1021/jacs.5b10119
