Aug. 12, 2016 Research Highlight Biology
Cellular tug-of-war
A novel mechanism controlling the dynamics between tubule cells sheds light on cellular adhesion
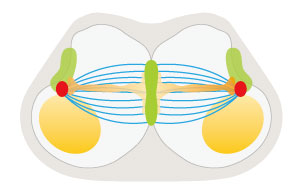 Figure 1: Schematic view of two migratory tubule cells making contact and beginning to pull toward opposite poles (green: transmembrane proteins at the cell adhesion interface; red: centrosome; blue: microtubule; orange: myosin; yellow: nucleus). Reproduced from Ref. 1 and licensed under CC BY 4.0 © 2016 K. Kato et al.
Figure 1: Schematic view of two migratory tubule cells making contact and beginning to pull toward opposite poles (green: transmembrane proteins at the cell adhesion interface; red: centrosome; blue: microtubule; orange: myosin; yellow: nucleus). Reproduced from Ref. 1 and licensed under CC BY 4.0 © 2016 K. Kato et al.
A surprising mechanism behind cellular coordination during tissue and organ development has been identified in a bioimaging study of fruit flies by RIKEN researchers1.
It is not at all clear how migratory tubule cells called tip cells find and identify their target tissues and establish connections with new cell surfaces during respiratory tubule development. During the formation of cellular junctions, a pair of cell tips, or ‘fusion cells’, establishes a new junction. This process involves dynamic cell shape change and highly coordinated membrane trafficking, which is also poorly understood.
To parse these cellular dynamics, Shigeo Hayashi of the RIKEN Center for Developmental Biology and his colleagues examined cell contractility and adhesion in the tracheal system—the complex network of tubes that delivers oxygen to the cells of an insect—of the fruit fly Drosophila.
They found that tracheal fusion cells orchestrate the migration of tracheal branch tubules by adhering and transforming into a donut shape through precise coordination of the paired fusion cells. “Conversion of spherical cells into a donut shape is an extremely rare shape change for animal cells,” Hayashi notes.
The team uncovered one of the mechanisms that give rise to this rare cell shape. Using various bioimaging and cell perturbation techniques, they discovered that the paired fusion cells undergo a highly balanced pulling event that involves the motor protein myosin applying a pulsed pulling force to the contact site (Fig. 1).
The researchers also found that the paired cell-to-cell adhesion complex in the cell interface is the target for microtubule association. The bundles of microtubules balance the pulling force. Consequently, when the microtubules are disrupted, the pulling force of each cell toward opposite poles is no longer balanced, and the fusion cells start a tug-of-war between paired cells.
In other words, the team discovered that the balance of cell contractility in the paired fusion cells is important for coordination, and they identified that the microtubule is a key coordinator of this force balance.
“We had anticipated that microtubules are needed for cell adhesion and fusion, and this was the case,” says Hayashi. “However, this tug-of-war-like contraction phenotype was unexpected. It was interesting to find that this extremely rare form of cell shape change is driven by the general intracellular machines (microtubules) in the configuration, similar to the mitotic spindle,” adds Hayashi.
The researchers next intend to examine the role of cell membrane fusion in the formation of these donut shapes.
Related contents
- Protein contortions keep cells together
- The chemical attraction underlying synaptic diversity
- Managing the body’s interior architecture
References
- 1. Kato, K., Dong, B., Wada, H., Tanaka-Matakatsu, M., Yagi, Y. & Hayashi, S. Microtubule-dependent balanced cell contraction and luminal-matrix modification accelerate epithelial tube fusion. Nature Communications 7, 11141 (2016). doi: 10.1038/ncomms11141
