Jan. 27, 2017 Research Highlight Chemistry
A both/and approach to creating polymers
A dual-mechanism way of forming polymers promises to lead to new polymers with unique structures
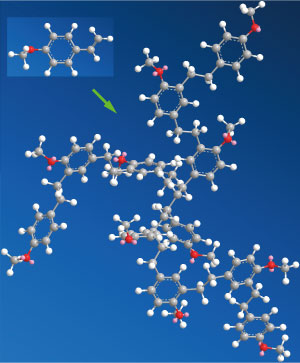 Figure 1: A polymer product containing multiple branches composed of alternating anisole and ethylene sequences formed by polymerization of para-methoxystyrene by activation of both carbon−carbon double bonds and carbon−hydrogen bonds (gray: carbon; white: hydrogen; red: oxygen). © 2017 Zhaomin Hou, RIKEN Center for Sustainable Resource Science
Figure 1: A polymer product containing multiple branches composed of alternating anisole and ethylene sequences formed by polymerization of para-methoxystyrene by activation of both carbon−carbon double bonds and carbon−hydrogen bonds (gray: carbon; white: hydrogen; red: oxygen). © 2017 Zhaomin Hou, RIKEN Center for Sustainable Resource Science
Polymers with unique structures that could be used in applications ranging from coatings to electronic devices have been produced by RIKEN researchers1. By combining two types of polymerization reactions in a single pot and using a rare-earth catalyst, the researchers produced polymers that are both highly branched and highly ordered.
Polymers are large molecules made by linking together lots of small ‘monomer’ molecules in a repeating pattern. For example, the everyday plastic polyethylene is formed by connecting small ethylene monomers (CH2=CH2) together in a chain.
There are two typical mechanisms by which monomers can join together. Some polymerization reactions proceed via a chain-growth mechanism in which monomers add to the growing polymer chain one by one, like threading beads on a string. Others proceed via a more chaotic step-growth mechanism in which monomer reacts with monomer and multiple short chains form, which gradually link together to form the final polymer.
Now, Zhaomin Hou from the RIKEN Center for Sustainable Resource Science and colleagues have developed a catalyst that has the rare ability of inducing certain monomers to undergo both chain-growth and step-growth polymerizations.
The team used a monomer called methoxystyrene, a molecule containing an ethylene group (a carbon–carbon double bond) and an anisole group (a methoxybenzene group). Until now, these monomers had only been shown to undergo chain-growth polymerization, connecting via their carbon–carbon double bonds. But Hou’s catalyst not only promotes chain-growth polymerization by activating the ethylene group, it also promotes step-growth polymerization by activating one of the carbon–hydrogen bonds of the anisole group.
“Cationic rare-earth metal alkyl complexes are highly active catalysts toward both C–H activation and C=C double-bond insertion,” Hou explains. “The special catalytic activity of these catalysts for this transformation springs from the strong affinity of rare-earth metal ions for oxygen, which facilitates the interaction between the catalyst and the anisole unit.”
The resulting polymers are unique, Hou adds. “The products contain multiple branches composed of perfectly alternating polar (anisole) and non-polar (ethylene) sequences (Fig. 1), which may find applications in areas such as coatings, polymer electrolytes and nanoscience.”
Having demonstrated the feasibility of combining chain-growth and step-growth polymerizations in one pot, the team is keen to explore other possible examples. “This work demonstrates that rare-earth metal-catalyzed C−H activation and C=C double bond insertion may provide unique opportunities for constructing novel polymer architectures that are difficult to make by other means,” says Hou. “We will continue to use this strategy to synthesize new functional polymers.”
Related contents
References
- 1. Shi, X., Nishiura, M. & Hou, Z. Simultaneous chain-growth and step-growth polymerization of methoxystyrenes by rare-earth catalysts. Angewandte Chemie International Edition 55, 14812-14817 (2016). doi: 10.1002/anie.201609065
