Oct. 13, 2017
Peering into blood vessel formation
Li-Kun Phng, Team Leader

Laboratory for Vascular Morphogenesis , RIKEN Center for Developmental Biology
Please describe your role at RIKEN.
I joined CDB as a team leader in October 2016 and am still in the process of hiring scientists and setting up a zebrafish study facility for my research.
What is your current research focus?
I’m primarily interested in understanding how the cells that line blood vessels, known as endothelial cells, behave and how they coordinate to form new blood vessels. We employ genetics, pharmacology and advanced microscopy to investigate the fundamental principles of blood vessel formation using the zebrafish as our model organism. We use zebrafish because we can easily label their cellular and subcellular structures with fluorescent proteins and their embryos are optically transparent. These properties are useful as we use live imaging to help us visualize the dynamics of blood vessel formation.
What are the potential applications?
Excessive or abnormal blood vessel formation can cause diseases such as cancer metastasis, when cancer cells spread to different parts of the body through the vessels, and diabetic retinopathy, where excessive blood vessel formation in the retina can lead to blindness. I hope that my work on the basic mechanisms of vascular development will lead to a better understanding of vascular diseases, which will enhance treatments.
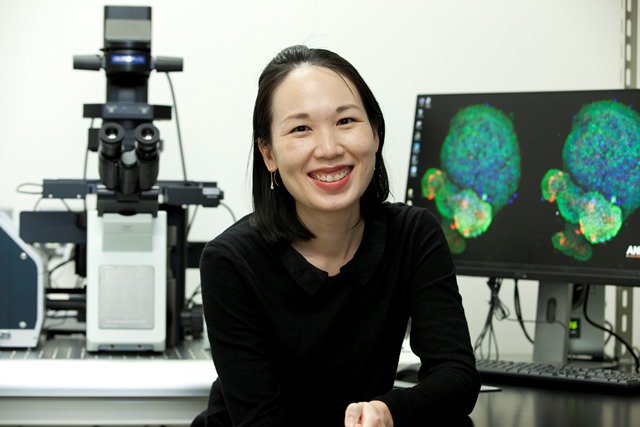
What excites you the most about your current research?
My research entails a substantial amount of live imaging and it’s very exciting to be able to observe the dynamics of cellular processes first-hand, particularly when we observe new behaviors and phenomenon.
What do you think has been the most interesting discovery in your field in the last few years? How has it influenced your research?
There is a growing body of evidence on the importance of hemodynamic forces (i.e. blood flow and pressure) in regulating endothelial cell behavior and blood vessel formation. I was part of a recent discovery published in Nature Cell Biology that identified a new type of membrane protrusion—called inverse blebbing—at the apical membrane of endothelial cells. These inverse blebs drive the expansion of the lumen, which is a structure that allows blood to flow through the vessels. This process is therefore important in the formation of functional blood vessels.
How has being at RIKEN helped your research?
I’ve had a lot of support from CDB to set up my lab. I am also glad that there is child support to help working parents—particularly the nearby affiliated childcare center where I send my daughter when I’m at work.
Please tell us about your professional and personal goals.
My long-term goal is to understand how upstream signals and hemodynamic forces regulate endothelial cell behavior to shape blood vessel architecture. To achieve this, I believe that interdisciplinary approaches are required, and I would like to forge collaborations with scientists from different backgrounds—such as physics and engineering—to understand the mechanics behind building blood vessels.
