Feb. 5, 2014
A new optical microscope enabling deep imaging of biological samples
RIKEN No.: 07895
Inventors
Keisuke Isobe and Katsumi Midorikawa (Laser Technology Laboratory)
Background
Nonlinear optical microscopes utilizing near infrared light is optimal for making observation on deep parts of biological samples in which light tends to scatter. Normally, only the area around the focusing point can be observed as signal light is generated around at the focus using the objective lens, and tomogram images are taken by moving this focusing point.
However, as the observed part becomes deeper, optical loss grows due to absorption or scattering and light generated at areas other than the focusing point (background light) acts as noise, making observation difficult, since nonlinear phenomenon is induced not only at the focusing point but also on the sample surface. Furthermore, spatial resolution is reduced in principle, due to reasons including the use of near infrared light which has approximately twice the wavelength of visible light and use of it at a low effective numerical aperture.
Summary
The intensity of signal light generated by nonlinear 2-wavelength excitation optical processes such as 2-photon absorption, 2-photon fluorescence, sum-frequency generation, difference frequency generation and stimulated Raman scattering is proportional to the product of the intensities of the 2 wavelength pulses, and is induced by the temporal and spatial overlapping of the 2 wavelength pulses. By modulating the spatial overlap of 2 wavelength pulses, it is possible to induce intensity modulation in the signal light generated. This intensity modulation reaches largest on the focal plane in the optical axis direction and becomes smaller as it is farther away from the focal plane. In addition, the frequency characteristics of the modulated signal light intensity vary as the position shifts from the center of the focal spot toward the outer areas.
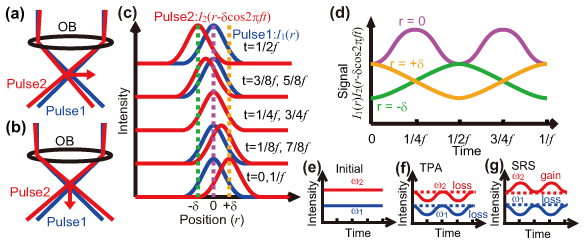
Fig.1: Principle of SPOMNOM
The new microscopy "SPOMNOM" uses 2 wavelength pulse lasers as the light sources, with one optical pulse laser fixed and the other given relative position modulation at a specified frequency so that intensity modulation is induced only in the signal light generated around the focusing point and the generated signal light is lock-in detected with modulation frequency. It enables suppression of the background signal generated in areas other than the focusing point (background light) and observation of deeper parts. Furthermore, spatial resolution is improved as the detected signal is generated from an area smaller than the focal spot.
This "SPOMNOM" method can be applied to all conventional nonlinear optical microscopes. Therefore, it is possible to simultaneously make observations using various nonlinear optical phenomena such as sum-frequency generation to observe asymmetrical molecules, 4-wave mixing to observe the refractive index, stimulated Raman scattering to observe the molecular vibration conditions in addition to nondegenerate 2-photon fluorescence to observe the fluorescent molecules.
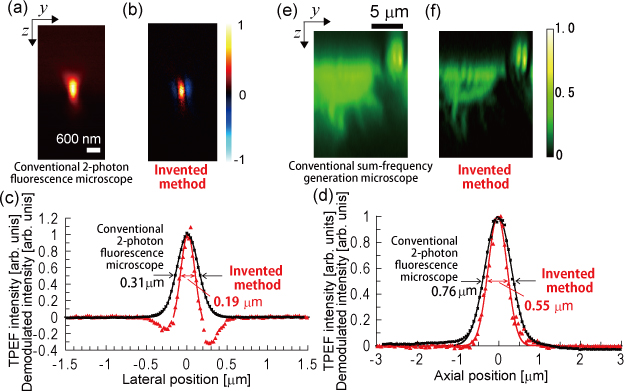
Fig.2: Improvement in spatial resolution
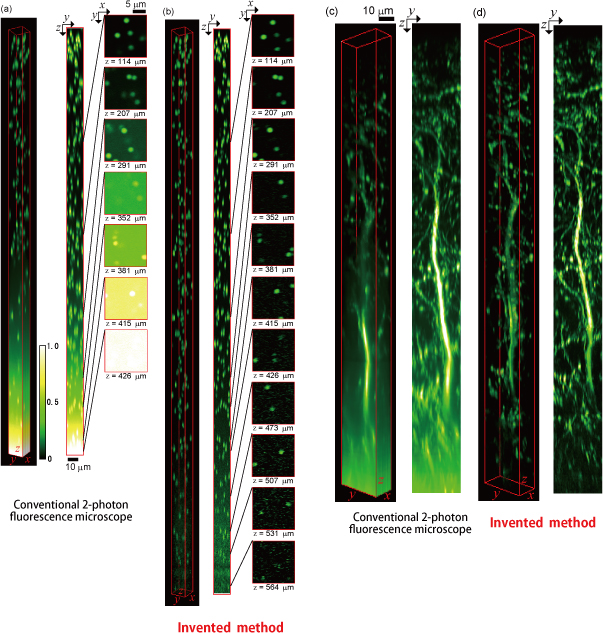
Fig.3: Improvement in deep imaging capability
Merits
- Background light generated near the surface can be eliminated (suppressed to approximately 1/100 of the conventional level).
- Observable depth improved to twice that of the conventional method
- Spatial resolution improved to 1.4- to 2-fold when the same numerical aperture is used
- Use with high numerical aperture possible (spatial resolution 200 nm or lower)
Applications
- Research in biology, medicine and drug development (such as visualization of the immune effect of lymphatic tissue, and visualization of brain tissue functions)
- Pathological examination conducted by cutting out tissue sections and so forth (it might be possible to address methods that require no staining or sections to be cut out.)
- Nondestructive inspection of semiconductors, optical devices and so forth
- Nondestructive inspection of food items
References
- 1.PCT/JP2012/052377
- 2.K. Isobe et al., Biomed. Opt. Express, 3, 1594-1608 (2012).
- 3.K. Isobe et al., Biomed. Opt. Express, 4, 1548-1558 (2013).
- 4.K. Isobe et al., Biomed. Opt. Express, 4, 1937-1945 (2013).
