May 28, 2010 Press Release Biology Chemistry
Finding the needle in the proteomic haystack
Researchers at the RIKEN Advanced Science Institute have demonstrated that a technique for tagging and profiling proteins can be used to accurately classify anti-cancer drugs based on the molecules they target. The technique, developed in collaboration with a researcher at the University of Tsukuba, promises to accelerate the process of proteomic profiling and contribute to more effective drug discovery.
While essential for effective treatment, our understanding of the complex relationship between drugs and their molecular targets is far from complete. Drug efficacy is thus low, and drugs are often accompanied by dangerous side effects. Targeted cancer therapies, which act by interfering with specific molecules contributing to tumor growth, can bring therapeutic benefits to certain patients, yet are not free of these problems. Solving them depends on connecting drugs to their molecular targets, a task complicated by the huge variety of proteins and complexity of drug-target networks.
In recent years, the field of proteomics has produced powerful techniques that simplify this task by accurately identifying proteins and their interactions. The research team used one of these techniques, two-dimensional difference gel electrophoresis (2D-DIGE), to analyze and compare changes of protein expression levels in HeLa cancer cells treated with 19 well-known anti-cancer agents. The method successfully classified compounds of different structures but sharing molecular target(s), while revealing that compounds previously reported to inhibit the same process in fact were clustered into different groups, highlighting differences in underlying interaction mechanisms.
Described in the journal Chemistry and Biology, the findings broaden the scope of proteomic profiling by enabling researchers to characterize specific compounds in terms of how they interact with biomolecules. While refining the drug discovery process, this advance also contributes to minimizing side-effects, potentially supporting the future development of safer and more effective anticancer drugs.
Contact
Hiroyuki Osada
Makoto Muroi
Chemical Library Validation Team
RIKEN Advanced Science Institute (ASI)
Tel: +81-(0)48-467-4839 / Fax: +81-(0)48-462-4669
Jens Wilkinson
RIKEN Global Relations and Research Coordination Office
Tel: +81-(0)48-462-1225 / Fax: +81-(0)48-463-3687
Email: pr@riken.jp
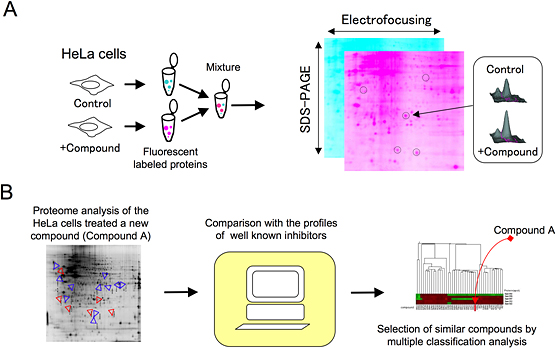
Figure 1: Proteome analysis by 2D-DIGE (2 Dimensional Fluorescence Difference Gel Electrophoresis) and classification of compounds
(A) Lysates of HeLa cells treated with a compound or control cells are labeled with a different fluorescent dye. After developing the mixture of the lysates by 2D-electrophoresis, the spots of proteins on the gel are quantitated by the 2D-DIGE system. The quantification is performed in one gel that provides reproducible data.
(B) Proteome analysis of HeLa cells treated with a new candidate compound whose target is unknown. To compare the 2D-DIGE proteome analysis data with those of well-characterized compounds, multiple classification analysis is carried out and similar compounds are selected. The target of the new compound is then verified by analyzing the interaction of the target molecule of similar compounds.
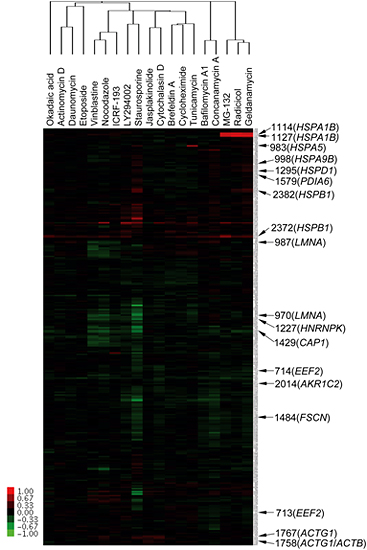
Figure 2: Clustering of 19 well-characterized compounds
After treatment of HeLa cells with 19 well-characterized compounds, proteome analysis of each lysate was performed using the 2D-DIGE system and then subjected to hierarchical cluster analysis resulting in the construction of a tree diagram. Compounds sharing a common target, such as HSP90 inhibitors (geldanamycin and radicicol), tubulin inhibitors (nocodazole and vinblastine) and V-ATPase inhibitors (bafilomycin A1 and concanamycin A), were classified the same tree.
