Jul. 12, 2010 Press Release Biology
Optogenetic probes to image brain electrical activity in laboratory mice
RIKEN scientists have developed a genetically-encoded fluorescent sensor that can act as a direct optical indicator of signaling activity in the living brain. This powerful tool, described in a new article from Nature Methods, promises to help neuroscientists identify and monitor the behavior of neural circuits involved in motor activity, sensory perception and other functions.
Researchers have historically analyzed the complex electrical activity of the brain using an invasive approach involving tiny electrodes, whose large size relative to individual nerve cells has limited the number of locations from which neuronal activity can be sampled. Optimal imaging methods overcome this limitation with molecular size probes that transform the electrical signals into an optical reporter signal.
The voltage-sensitive fluorescent proteins (VSFPs) developed by Thomas Knöpfel's team at the RIKEN Brain Science Institute represent an important step in this direction. These are engineered proteins that reside within the membranes of neurons, each of which is fused to two different fluorescent proteins. Whenever a neuron receives a stimulatory signal, the resulting change in voltage potential in the cell membrane causes the VSFPs to rearrange into a configuration that causes a readily detectable change in the optical signal generated by the VSFP, in a phenomenon known as Förster Resonance Energy Transfer (Figure 1).
Knöpfel's laboratory pionered the development of these sensors for more than 10 years but up to now the function of these probes was only demonstrated by recording electrical activity from 2-dimensional networks of cultured nerve cells. In the latest edition of Nature Methods, the team presents the first experimental confirmation that these probes are able to report electrical activity of nerve cells in the brains of living mice. The researchers used genetically modified mice to localize the VSFP probe within specific subsets of cortical neurons within a brain area called the somatosensory cortex. Each mouse whisker is wired to discrete neural circuits in the somatosensory cortex, and the researchers found that they could readily detect changes in the membrane voltage of these circuit elements as each whisker was manipulated. Based on these experiments, they were essentially able to reconstruct maps of the cell populations that operate as 'receptive areas' for individual whiskers (Figure 2).
Being genetically encoded, VSFPs offer several advantages over other commonly-used approaches to monitoring neuronal activity. They can essentially be 'programmed' for selective expression within specific subtypes of neurons or particular regions of the brain, and could be used to chart long-range neural circuits extending over considerable distances, unlike fluorescent dyes that label cells non-specifically and can only be applied within a relatively limited volume of the brain. Other genetically-encoded sensors have been developed that respond to calcium flux in the immediate aftermath of neuronal firing, but these represent indirect indicators and generally respond more slowly to neuronal activity.
Given the high degree of spatial and temporal resolution displayed by the VSFPs in this study, Knöpfel is confident that they will prove a useful tool for researchers hoping to understand how patterns of neuronal activity correlate with behavior or physiological changes in the living brain. "The ability of VSFPs to report faster signals, along with genetic targeting, will allow new approaches to the study of the dynamic interaction of assemblies of neurons," he says. "This will facilitate the investigation of fundamental questions of information processing in the brain, such as the circuit operations involved in sensing our environment and generation of body movements, but will also be applicable to directly visualize cognitive functions."
Contact
Thomas Knöpfel
Laboratory for Neuronal Circuit Dynamics
RIKEN Brain Science Institute
Tel: +81-(0)48-467-9740 / Fax: +81-(0)48-467-9739
Jens Wilkinson
RIKEN Global Relations and Research Coordination Office
Tel: +81-(0)48-462-1225 / Fax: +81-(0)48-463-3687
Email: pr@riken.jp
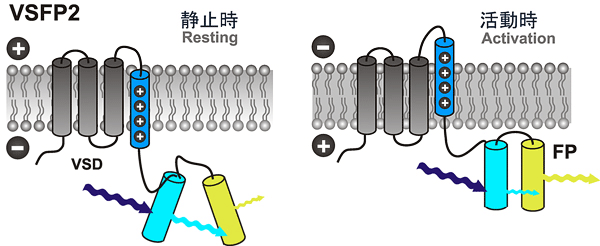
Figure 1: VSFP 2.3/2.42 mechanism
When in their resting state, nerve cells excite a bluish-purple light at a wavelength of 440 nm, while in an excited state they emit blue light with a wavelength between 470 and 495 nm. When neural activity triggers a change in the membrane potential, the spatial structure of the VSFP2.3/2.42 changes as well, altering the direction of the two fluorescence proteins. This triggers the phenomenon of Fluorescence Resonance Energy Transfer (FRET), in which fluorescence excitation energy is transferred through resonance, resulting in an increased emission of yellow fluorescence. Changes in potential can thus be observed.
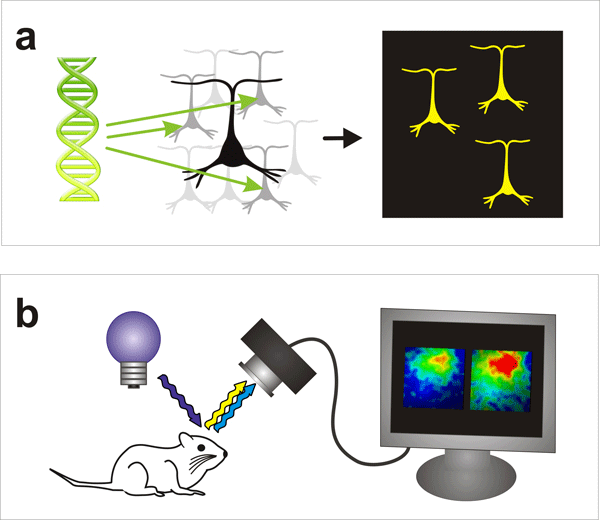
Figure 2: Visualization of activity of neuronal cell population
(a) Voltage-sensitive fluorescent proteins are expressed in nerve cells. When genes producing VSFPs are injected into cells (left) and they are exposed to fluorescent light, those cells emit a specific color (right, yellow).
(b) Monitoring neural activity using VSFP. The brain of a mouse with specific nerve cells containing VSFPs is exposed to light at a given wavelength. When the membrane potential changes with neural activity, the relative intensity of fluorescence observed also changes, and thus one can observe electrical activity in real-time.
