Nov. 18, 2022 Press Release Medicine / Disease
Inducing hibernation-like state in mice can protect organs during heart surgery
Researchers led by Hidetoshi Masumoto and Genshiro Sunagawa at the RIKEN Center for Biosystems Dynamics Research (BDR) in Japan have developed a new method of protecting organs during heart and aortic surgery when blood circulation has to be blocked. Rather than relying on cold temperatures to induce hypometabolism and reduce the need for oxygen, the technique works by stimulating Q neurons in the brain, which slow metabolism down to a hibernation-like state. In this proof-of-concept study, the procedure protected mouse kidneys from damage due to lack of oxygen and avoided harmful side effects related to extended hypothermia. The findings could lead to new ways of performing similar surgeries in people.
For some kinds of heart and aortic surgery, doctors must cut off blood circulation as they work to repair the aorta. For the last 50 years, this type of surgery has been performed after using cold temperatures to induce deep hypothermia, which slows down the body’s metabolism so that organs can survive with very little oxygen. While this is effective in protecting organs like the kidneys, it also reduces blood coagulation, causing excessive bleeding and the need for blood transfusions.
Masumoto and his team at RIKEN BDR have been looking for ways to slow down metabolism without inducing hypothermia. We know that many animals—such as bears and squirrels—can hibernate, and that despite the extremely slow metabolism during hibernation, they are healthy when they wake up. However, like most animals, people do not hibernate. This was the end of the story until a few years ago when Sunagawa’s group discovered a way to induce a hibernation-like state in mice—animals that do not normally hibernate. “If we can induce them, there are many possibilities for using hibernation-like states in cardiovascular medicine, resuscitation medicine, or other cases in which organ protection via hypothermia is insufficient or inappropriate,” says Sunagawa.
But first, the technique’s effectiveness needs to be verified in animal models. The new study is the first clinical implementation of the technique, and tested its effectiveness using a mouse model of aortic surgery that requires circulatory arrest. The team’s previous studies showed that hibernation-level hypometabolism could be achieved by activating special neurons in the brain’s hypothalamus called Q neurons. Chemogenetic biotechnology allowed the researchers to trigger activation of these specific neurons with an injection. They compared four groups of model mice that varied in whether or not cold temperatures were used to induce hypometabolism and whether or not Q neurons were activated.
Kidney damage and renal function were evaluated by looking at levels of biomarkers in the blood. Analysis showed hypometabolism induced by Q-neurons at normal temperatures protected the kidneys as well as that induced using hypothermia. “With these results, we now know that Q neurons ¬induced hibernation-like states can be used to protect organs,” says Masumoto.
The ultimate goal of Masumoto and Sunagawa’s research is to slow down people’s metabolism during heart surgery, or for other medical reasons, using some variation of this technique. But because Q neurons in people cannot be selectively activated the way they can in experimentally engineered mice, the team is now looking at ways to protect organs downstream from the brain. “Activating Q neurons triggers some sequence of biological events that allows organs to exist in a hypometabolic state for days,” explains Sunagawa. “Once we know precisely what these events are, we are confident we can induce them pharmacologically in the body, without needing to first activate the Q neurons.”
The study was published in JTCVS Open on November 7.
Rate this article
Reference
Contact
Hidetoshi Masumoto, Senior Scientist
Laboratory for Organismal Patterning
RIKEN Center for Biosystems Dynamics Research
Genshiro A. Sunagawa, Team Leader
Laboratory for Hibernation Biology
RIKEN Center for Biosystems Dynamics Research
Adam Phillips
RIKEN International Affairs Division
Tel: +81-(0)48-462-1225
Email: adam.phillips [at] riken.jp
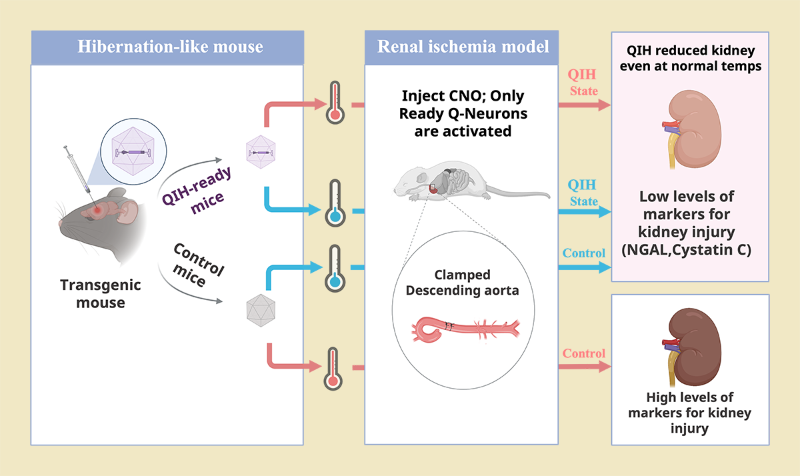
Summary of experiment:
All mice were then injected with clozapine-N-oxide, which activated “ready” Q-neurons. At the same time, the descending aorta was clamped, mimicking circulatory arrest. This procedure was done at normal temperatures or cold temperatures, and in “Q-neuron ready” and control mice. At normal temperatures, the control mice did not go into a hypometabolic state and the kidneys showed signs of extensive damage. In contrast. at normal temperatures, the mice with activated Q-neurons did go into a hypometabolic state and their kidneys shows much less damage.
