Jun. 30, 2017 Research Highlight Biology
A golden opportunity for drug targeting
Tissue-targeting gold complexes could be used to activate drugs only at specific sites in the body
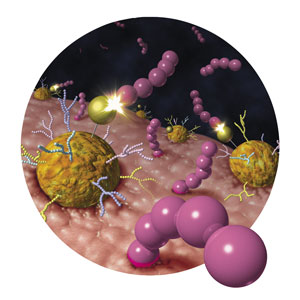 Figure 1: Gold complexes can speed chemical reactions at targeted sites in the body. © 2017 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
Figure 1: Gold complexes can speed chemical reactions at targeted sites in the body. © 2017 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
Gold complexes can be delivered to target organs in living mice, where they can speed chemical reactions for diagnostic or therapeutic purposes, show RIKEN researchers1. This paves the way for future clinical applications in humans.
“Our method enables various therapeutic molecules to be synthesized directly at the target organs in living organisms,” says Katsunori Tanaka, chief scientist of the RIKEN Biofunctional Synthetic Chemistry Laboratory. This ability to synthesize diagnostic or therapeutic molecules exactly where they are needed can reduce the amount of compound needed and the risk of side effects.
Most previous demonstrations of metal-mediated reactions in biological systems involved human cells in a dish, while a few studies have been done in bacteria or developing fish embryos. But for the method to be developed for therapy, it is vital to show that it works with tissue specificity in mature mammals.
To achieve this, Tanaka’s team linked a gold-ion catalyst via an intermediate locking system to a protein called albumin, which can be tagged with sugar molecules known as glycans. These sugary markers determine where the complex will go when it is injected into the body—attach one kind of glycan, and the complex will go to the liver; attach another, and it will head to the intestine.
 Katsunori Tanaka and his team have developed a method that allows drugs to be synthesized directly at target organs in living animals. © 2017 RIKEN Biofunctional Synthetic Chemistry Laboratory
Katsunori Tanaka and his team have developed a method that allows drugs to be synthesized directly at target organs in living animals. © 2017 RIKEN Biofunctional Synthetic Chemistry Laboratory
The team injected their gold complexes into mice and then injected an imaging agent. The agent fluoresces only when gold complexes catalyze the formation of amide bonds between the imaging agent and proteins on the surface of organs. By imaging where the mice glowed, the researchers showed that their gold complexes arrived at the target organ, the liver or intestine, within just a couple of hours after injection. The same basic idea, Tanaka explains, could work with a drug instead of an imaging probe.
The researchers have also recently developed a way of engineering glycan complexes that include more than one type of sugar molecule2. This increased the precision of tissue targeting, explains Tanaka. “We can even target specific tumor cells out of many other tumor cells, which cannot be realized by other methods,” he says.
Pharmaceutical companies have abandoned many once-promising drug candidates either because they showed toxic side effects in off-target tissues or because the drugs were too unstable be delivered intact to the desired organ. Using gold to activate drugs at their desired site “could re-optimize molecules that have been dropped during drug development,” Tanaka says.
Tanaka’s team is now trying to adapt the technique for use in rabbits and monkeys—and, soon, he hopes, in humans too.
Related contents
- Glycocluster design could lead to targeted drug delivery
- Blocking obesity with a protein-sugar combination
References
- 1. Tsubokura, K., Vong, K. K. H., Pradipta, A. R., Ogura, A., Urano, S., Tahara, T., Nozaki, S., Onoe, H., Nakao, Y., Sibgatullina, R. et al. In vivo gold complex catalysis within live mice. Angewandte Chemie International Edition 56, 3579–3584 (2017). doi: 10.1002/anie.201610273
- 2. Latypova, L., Sibgatullina, R., Ogura, A., Fujiki, K., Khabibrakhmanova, A., Tahara, T., Nozaki, S., Urano, S., Tsubokura, K., Onoe, H. et al. Sequential double “clicks” toward structurally well-defined heterogeneous N-glycoclusters: The importance of cluster heterogeneity on pattern recognition in vivo. Advanced Science 4, 1600394 (2017). doi: 10.1002/advs.201600394
