Sep. 22, 2021 Perspectives Biology
Harnessing natural killer T cells to stop cancer
Innovative use of stem cell technology to ramp up production of natural killer T cells could hold the key to improved cancer treatments.
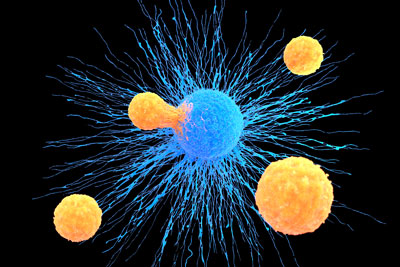 Natural killer T (NKT) cells are a subset of T cells (orange) that coexpress an αβ T-cell receptor and express a variety of molecular markers that are typically associated with NK cells. Among their many roles they can induce the death of cancers cells (blue), making NKT cells a fresh target for therapeutic development. © ROGER HARRIS / SCIENCE PHOTO LIBRARY / GETTY IMAGES
Natural killer T (NKT) cells are a subset of T cells (orange) that coexpress an αβ T-cell receptor and express a variety of molecular markers that are typically associated with NK cells. Among their many roles they can induce the death of cancers cells (blue), making NKT cells a fresh target for therapeutic development. © ROGER HARRIS / SCIENCE PHOTO LIBRARY / GETTY IMAGES
Cancer accounted for nearly 10 million deaths worldwide in 2020 and is the leading cause of death in Japan1. Immunotherapy primes the patient’s immune system to fight cancer cells and offers a promising alternative to the usual treatments of chemotherapy, radiation therapy and surgery. Researchers at the RIKEN Center for Integrative Medical Sciences (IMS) are investigating a number of immunology and cancer therapies, and our team at the RIKEN IMS Laboratory for Developmental Genetics is spearheading efforts to develop new cancer immunotherapy strategies by harnessing the power of natural killer T (NKT) cells.
In September 2020, we started recruiting for the world’s first clinical trial using NKT cells derived from induced pluripotent stem (iPS) cells to treat patients with head and neck cancer. This followed more than a decade of intensive basic research and validation studies with colleagues at Chiba University. Four to 18 patients will be enrolled in the trial and may undergo the novel treatment over the next two years. While it is too soon to evaluate efficacy, confirming the safety of iPS-derived NKT cell therapy will be a key step toward the development of this potentially groundbreaking treatment for many different types of cancer.
Roots at RIKEN
 Since late 2020, a clinical trial designed to suppress tumors using iPS-derived NKT cells has been conducted on Japanese patients with squamous cell carcinoma (pictured), a condition accounting for 80–90% of all head and neck tumors. © Science History Images / Alamy Stock Photo
Since late 2020, a clinical trial designed to suppress tumors using iPS-derived NKT cells has been conducted on Japanese patients with squamous cell carcinoma (pictured), a condition accounting for 80–90% of all head and neck tumors. © Science History Images / Alamy Stock Photo
The unique invariant antigen receptor (Vα14Jα18 in mice) of NKT cells was discovered in 19862 by immunologist Masaru Taniguchi, then at Chiba University and now senior visiting scientist at RIKEN IMS. His work spurred research into the role of NKT cells in the regulation of immune responses, opening up the development of anticancer therapeutics. The discovery of NKT cells has been designated as a ‘Pillar of Immunology’ by the American Association of Immunologists3.
Because of their unique receptors, NKT cells recognize lipid antigens presented by the cell surface molecule monomorphic CD1d. They rapidly produce large amounts of cytokines (such as interferon-gamma, IFN-γ) which are critical to activating antitumor functions4,5. Studies of NKT cell-deficient mice have demonstrated6 that NKT cells play an essential role in triggering antitumor immune responses and establishing immunological memory against tumor cells.
With a view to unlocking the potential of these rare cells as a cancer therapy, the search was on for a suitable molecule to trigger NKT cells7. Taniguchi and his team succeeded in singling out alpha-galactosylceramide (α-GalCer), a glycolipid derived from the sea sponge Agelas mauritianus, in 1997. They showed that α-GalCer binds to CD1d. The resulting complex leads to enhanced NKT cell activity and high levels of cytokine production, which mediates adjuvant activity and activates various other antitumor immune cells.
A preclinical breakthrough came in 1999 with intravenous administration of α-GalCer to mice with liver cancer8. The treatment, involving the use of dendritic cells pulsed with α-GalCer, resulted in complete inhibition of melanoma metastasis within just one week.
Subsequent studies have shown that after stimulation with α-GalCer, NKT cells appear to activate both CD8+ cytotoxic T cells (to destroy major histocompatibility complex (MHC)-positive tumor cells) and natural killer cells (to eliminate MHC-negative tumor cells)9,10. As tumors frequently contain both MHC-positive and -negative cell types, this remarkable ability to attack cancer cells on two fronts has led researchers to hail NKT cells as “an ideal anti-tumor immunotherapeutic”11 that could potentially lead to “complete eradication of tumors without relapse”12.
In 2001, a series of studies to assess the effects and safety of α-GalCer-pulsed dendritic cell therapy began at Chiba University Hospital, led by Shinichiro Motohashi13. A clinical trial of NKT cell-targeted immunotherapy using α-GalCer for patients with advanced non-small cell lung cancer began in March 2004. A cohort of 17 patients showed increased IFN-γ production and had a median survival time of 18.6 months,14 which is considerably longer than the typical survival time of less than 12 months. The results also suggested a mechanism by which NKT cells are able to induce long-term immune memory.
While α-GalCer treatment continues to be developed and explored, both in Japan and overseas, the stark fact remains that many cancer patients are ineligible for this treatment because their NKT cell count is too low. Even in the blood of healthy individuals, NKT cells comprise only 0.1% of total lymphocytes. This makes it extremely challenging to harvest NKT cells from the body and culture them to numbers sufficient for clinical use.
To overcome this problem, a multidisciplinary team at RIKEN developed a technique to generate large quantities of NKT cells using induced pluripotent stem (iPS) cells in mice in 201015. They succeeded in generating millions of functionally competent iPS-derived NKT cells, which secreted high levels of IFN-γ and suppressed tumor growth in live mice.
Following proof of concept16 for clinical application of human iPS-derived NKT cells in 2016, a clinical trial began at Chiba University Hospital in late 2020. iPS cells were made from NKT cells collected from the blood of healthy donors and cultured into large numbers. These iPS cells were then redifferentiated to NKT cells and injected into patients. Each patient received around 50 million iPS-derived NKT cells in one injection, followed by two further injections spaced two weeks apart.
The focus of the study is on patients with squamous cell carcinoma (a condition accounting for 80–90% of all head and neck tumors) who have previously not responded to surgery, radiation therapy, chemotherapy or a combination of these therapies. Those with cachexia (a wasting disease seen in many end-stage cancer patients) were excluded from the study, which limits participant recruitment.
The most important investigation is safety. The two biggest risk factors are iPS cells becoming cancerous themselves and patients exhibiting allogeneic responses (rejecting immunologically incompatible cells created from another person).
Provided that the preliminary results are satisfactory, a combination therapy of α-GalCer-pulsed dendritic cell treatment and iPS-derived NKT cells may be explored for tumor suppression. We will see results in 2022 at the earliest.
Future prospects
Devising new ways to make iPS-derived NKT cells more active presents an ongoing challenge.
Human leukocyte antigen (HLA) molecules mediate allogeneic rejection, but HLA can be knocked out at the iPS phase by using technologies such as CRISPR-mediated editing.
This process could also extend the retention period of iPS-derived NKT cells in the body. Efforts are also underway to make iPS-derived NKT cells more immunogenic through the application of chimera antigen receptor (CAR) T-cell therapy. This involves genetically engineering iPS-derived NKT cells to express a synthetic receptor that can bind tumor-specific antigens. As CD19-specific CARs are already successfully used in the treatment of patients with B cell-derived leukemia17, this is another promising avenue of research.
In order to scale up, there is a need for a simpler, more optimized protocol. It currently takes 40–50 days to generate sufficient numbers of NKT cells for treatment. However, based on evidence that NKT cells appear in mice on day 11 after fertilization, this could be shortened to as little as 2–3 weeks. A long-term goal will be to develop a cocktail of differentiation factors to facilitate the industrial generation of iPS-derived NKT cells, which would make the cells more cost effective.
Before such dreams can be realized, there is still a great deal of basic research to be done. There is much scope for informatics and data scientists to help us trace the developmental trajectory of iPS cells to NKT cells. Shin-ichiro Fujii, team leader at the RIKEN IMS Laboratory for Immunotherapy, is working on artificial adjuvant vector cell therapy, another variation of NKT cell-targeted therapy, for the treatment of relapse and refractory acute myeloid leukemia. In addition to the fruitful collaboration with Motohashi’s group at Chiba University, there may be more collaborations with other university hospitals nationwide and around the globe.
References
- 1. Ferlay, J., Ervik, M., Lam, F., Colombet, M., Mery, L. et al. Global Cancer Observatory: Cancer Today. Lyon: International Agency for Research on Cancer (2020). Available from: https://gco.iarc.fr/today/home Accessed June 2021.
- 2. Imai, K., Kanno, M., Kimoto, H., Shigemoto, K., Yamamoto, S. et al. Sequence and expression of transcripts of the T-cell antigen receptor α-chain gene in a functional, antigen-specific suppressor-T-cell hybridoma. Proceedings of the National Academy of Sciences USA 83, 8708–8712 (1986). doi: 10.1073/pnas.83.22.8708
- 3. Koseki, H., Imai, K., Nakayama, F., Sado, T., Moriwaki, K. et al. Pillars article: Homogenous junctional sequence of the V14+ T-cell antigen receptor α chain expanded in unprimed mice. The Journal of Immunology 193 (3), 993-997 (2014). https://www.jimmunol.org/content/193/3/993
- 4. Arase H., Arase N., Nakagawa K., Good, R.A. & Onoe, K. NK1.1+ CD4+ CD8− thymocytes with specific lymphokine secretion. European Journal of Immunology 23, 307–310 (1993). doi: 10.1002/eji.1830230151
- 5. Yoshimoto, T. & Paul, W.E. CD4pos, NK1.1pos T cells promptly produce interleukin 4 in response to in vivo challenge with anti-CD3. Journal of Experimental Medicine 179, 1285–1295 (1994). doi: 10.1084/jem.179.4.1285
- 6. Cui, J., Shin, T., Kawano, T., Sato, H., Kondo, E. et al. Requirement for Vα14 NKT cells in IL-12-mediated rejection of tumors. Science 278 (5343) , 1623–1626 (1997). doi: 10.1126/science.278.5343.1623
- 7. Kawano, T., Cui, J., Koezuka, Y., Toura, I., Kaneko, Y. et al. CD1d-restricted and TCR-mediated activation of Vα14 NKT cells by glycosylceramides. Science 278, 1626–1629 (1997). doi: 10.1126/science.278.5343.1626
- 8. Toura, I., Kawano, T., Akutsu, Y., Nakayama, T., Ochiai, T. et al. Cutting edge: inhibition of experimental tumor metastasis by dendritic cells pulsed with α-galactosylceramide. The Journal of Immunology 163, 2387–2391 (1999). https://www.jimmunol.org/content/163/5/2387
- 9. Fujii, S., Shimizu, K., Smith, C., Bonifaz, L. & Steinman, R. M. Activation of natural killer T cells by α-galactosylceramide rapidly induces the full maturation of dendritic cells in vivo and thereby acts as an adjuvant for combined CD4 and CD8 T cell immunity to a co-administered protein. Journal of Experimental Medicine 198 (2), 267–279 (2003). doi: 10.1084/jem.20030324
- 10. Hermans, I. F., Silk, J. D., Gileadi, U., Salio, M., Mathew, B. et al. NKT cells enhance CD4+ and CD8+ T cell responses to soluble antigen in vivo through direct interaction with dendritic cells. The Journal of Immunology 171 (10), 5140–5147 (2003). doi: 10.4049/jimmunol.171.10.5140
- 11. Fujii, S., Shimizu, K., Okamoto, Y., Kunii, N., Nakayama, T. et al. NKT cells as an ideal anti-tumor immunotherapeutic. Frontiers in Immunology 4, 409 (2013). doi: 10.3389/fimmu.2013.00409
- 12. Taniguchi, M., Harada, M., Dashtsoodol, N. & Kojo, S. Discovery of NKT cells and development of NKT cell-targeted anti-tumor immunotherapy. Proceedings of the Japan Academy, Series B Physical and Biological Sciences 91(7), 292–304 (2015). doi: 10.2183/pjab.91.292
- 13. Ishikawa, A., Motohashi, S. et al. A phase I study of a-galactosylceramide (KRN7000)-pulsed dendritic cells in patients with advanced and recurrent non-small cell lung cancer. Clinical Cancer Research 11, 1910–7 (2005). doi: 10.1158/1078-0432.CCR-04-1453
- 14. Motohashi, S., Nagato, K. et al. A phase I-II study of a-galactosylceramide-pulsed IL-2/GM-CSF-cultured peripheral blood mononuclear cells in patients with advanced and recurrent non-small cell lung cancer. The Journal of Immunology 182, 2492–2501 (2009). doi: 10.4049/jimmunol.0800126
- 15. Watarai, H., Fujii, S., Yamada, D., Rybouchkin, A., Sakata, S., Nagata, Y., Iida-Kobayashi, M., Sekine-Kondo, E., Shimizu, K., Shozaki, Y. et al. Murine induced pluripotent stem cells can be derived from and differentiate into natural killer T cells. The Journal of Clinical Investigation 120(7), 2610–2618 (2010). doi: 10.1172/JCI42027
- 16. Yamada, D., Iyoda, T., Vizcardo, R., Shimizu, K., Sato, Y., Endo, T. A. Kitahara, G., Okoshi, M., Kobayashi, M., Sakurai, M. et al. Efficient regeneration of human Vα24 + invariant natural killer T cells and their anti-tumor activity in vivo. Stem Cells 34(12) , 2852–2860 (2016). doi: 10.1002/stem.2465
- 17.17. Fousek, K., Watanabe, J., Joseph, S. K., George, A., An, X., Byrd, T. T., Morris, J. S., Luong, A., Martínez-Paniagua, M. A., Sanber, K. et al. CAR T-cells that target acute B-lineage leukemia irrespective of CD19 expression. Nature 35, 75–89 (2021). doi: 10.1038/s41375-020-0792-2
About the Researcher
Haruhiko Koseki, Deputy director, RIKEN Center for Integrative Medical Sciences

Haruhiko Koseki received his Doctor of Medicine and Ph.D. from Chiba University. He was a professor at the Graduate School of Medicine Department of Immunology at Chiba University between 1998 and 2004. He served as director of the Developmental Genetics Research Group at the RIKEN Research Center for Allergy and Immunology from 2001, where he studied the epigenetic regulation of Polycomb group genes in development. He serves as deputy director at the RIKEN Center for Integrative Medical Sciences and leads the Laboratory for Developmental Genetics.
