Oct. 20, 2023 Feature Highlight Medicine / Disease
Treating brain aneurysms before they burst
Studies reveal new potential drug targets to fix the problem of common, and potentially deadly, weakened arteries in the brain.
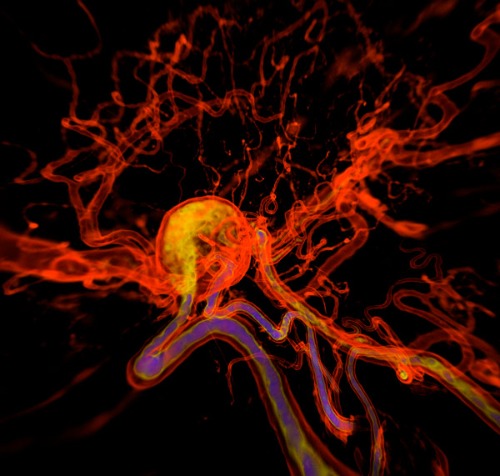
An intracranial aneurysm (round structure) is a section of an artery in the brain that starts to weaken and is at risk of bursting. © K H FUNG / SCIENCE PHOTO LIBRARY
Deadly danger lurks in the brain of one in 20 people. A section of an artery in the brain can start to weaken, ballooning into something called an intracranial aneurysm (IA) that is at risk of bursting.
As Hirofumi Nakatomi of the RIKEN Center for Brain Science explains, “The growth of an aneurysm increases the risk of its rupture, but there is currently no other treatment than surgery.” If an IA bursts, the bleeding often causes brain damage or death, even if a patient reaches a hospital quickly.
To find better treatments, scientists hope to unravel the biology behind these arterial bulges in the brain. Until recently, says Nakatomi, “the cause of intracranial aneurysms was largely unknown.” So researchers at RIKEN set out to find the answers.
A cluster of concern
While our genes account for about 10% of IAs—age, high blood pressure and alcohol consumption also increase the risk—Nakatomi wanted to dig deeper, especially into potential genetic causes. In a recent study1, he and his colleagues applied whole-exome sequencing of protein coding regions to samples from 65 human arteries, both with and without aneurysms.
“Unexpectedly, we found that more than 90% of the aneurysms had mutations in a common set of genes,” Nakatomi says.
In particular, tissues from cerebral aneurysms usually included mutations in a group of 16 genes in the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling pathway. This pathway is known to play a role in inflammatory responses and the formation of tumors.
Six of those genes also included mutations that change the platelet-derived growth factor receptor β (PDGFRβ), which is part of a protein family that transmits signals from the cell surface into the cell. Aberrations in PDGFRβ have also been linked to tumor cell proliferation, invasion, metastasis and development.
Many of the variants identified in the study arose from ‘missense’ mutations, which are changes in one base in a gene that swaps in a new amino acid. “These findings indicate that mutations are the major trigger of IA development in most cases,” Nakatomi says.
Although part of his ongoing work involves looking for more IA-related mutations, he doesn’t expect the list to grow. He says, “I am accumulating data on around 160 patients, and a second report could lead to a more precise percentage, but I am assuming the top two or three genes will be the same.”
Zeroing in on the problem
Beyond showing up in six of the IA-related genes, mutations in PDGFRβ appear in most of the weakened arteries. Importantly, IAs with a poor clinical prognosis (based on being deeper in the brain) included mutations in PDGFRβ in more than half of the cases of giant IAs.
In an effort to show the causal effect of PDGFRβ on IAs, the scientists created a mouse model of a rare IA, characterized by the invasion of cells (PDGFRB positive mural cells) into the wall of an artery, called an intracranial fusiform aneurysm (IFA).
“A variant of the PDGFRβ gene identified by our analysis was introduced into a mouse’s cerebral artery vessel wall with a viral vector,” Nakatomi says. The group then confirmed that 28 days after introduction of the mutant gene, aneurysm-like changes occurred in the main artery that supplies blood to the ‘inferior’ portion of the mouse brain around the brain stem. These changes were a spindle-like shape or ‘fusiform’ widening of the blood vessel, which is observed in human aneurysms.
Nakatomi’s team also revealed that PDGFRβ mutations appear most often in pericytes, which are cells on the outer membrane side of a blood vessel developing an aneurysm. Furthermore, they found that mutations in PDGFRβ triggered a process that can ultimately cause an enlargement of tissue hyperplasia. This was due to an increase in the reproduction of pericytes in the outermost ‘adventitial’ layers of capillaries first, then progressing into the inner media and endothelial layers.
At this point, Nakatomi and his colleagues started to think of ways that they could use this information to treat IAs. They started with a number of inhibitors that target tyrosine kinase receptors, a protein family transmits signals from a cell surface into the cell. These tyrosine kinase inhibitors have been developed for cancer therapy and Nakatomi’s group started by adding sunitinib—used to treat renal cancer—to human cells with a mutated PDGFRβ gene in a lab. They found that this inhibitor suppressed activity. The scientists also showed again that sunitinib suppressed the development of IAs in their IFA-like arterial expansion model in mouse populations.
Thus, they established for the first time a means of using genetic insertions to create a mouse model of cerebral aneurysm formation, and also a means of suppressing the their development in these models. But most excitingly, this finding indicates that intracranial aneurysms might one day be successfully treated with drugs, says Nakatomi.
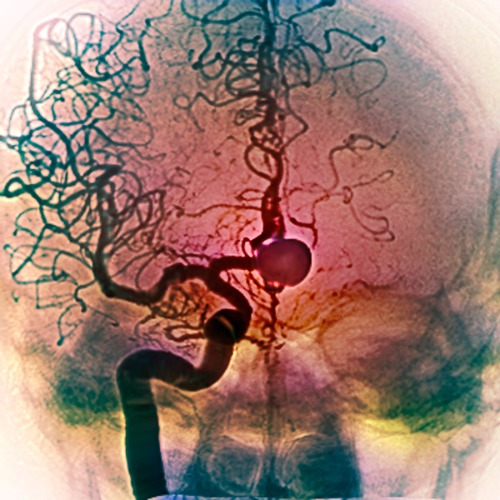
Researchers found that 90% of the aneurysms (pictured) they tested had mutations in a common set of genes. © SOVEREIGN / ISM / SCIENCE PHOTO LIBRARY
Finding more therapies
The team are now looking at other cancer drugs that might inhibit the development of IAs. “Many of the 16 genes that were frequently mutated in the cerebral aneurysm samples are cancer-related genes, some of which have already been identified as causes of solid tumors and for which molecular-targeted drugs and small-molecule compounds are being developed,” says Nakatomi.
Nonetheless, more research lies ahead. “If the inhibitory effect of sunitinib administration on aneurysm formation is verified in large mammals and then in human clinical trials, it could lead to the practical application of a completely new therapeutic agent for cerebral aneurysms,” he says.
Nakatomi and his colleagues also plan to explore the potential of targeting genes beyond PDGFRβ. They will start with AHNAK, the second most frequently detected mutated gene in IA samples.
If the scientists do come up with a drug treatment for IAs, the impact could be life changing for many. “We want to find an additional tool, one that prevents the need for surgery,” says Nakatomi, “and a drug would be almost as magical as a lightsaber.”
Rate this article
Reference
- 1. Shima, Y., Ota, S.S., Oyama, R., Tanaka, M., Kubota-Sakashita, M. et al. Increased PDGFRB and NF-κB signaling caused by highly prevalent somatic mutations in intracranial aneurysms. Science Translational Medicine 15(700), eabq7721 (2023). doi: 10.1126/scitranslmed.abq7721
About the researchers
Hirofumi Nakatomi

Hirofumi Nakatomi graduated from the University of Tokyo (UTokyo) School of Medicine in 1993. After a number of years as a practicing neurosurgeon in Japan, he returned to UTokyo to finish a PhD in Neurology in 1999. After this were stints at the Mayo Clinic and University of Cincinnati in the United States. Nakatomi returned to Japan to continue practicing and teaching. In 2018, he joined the Neurodynamic Medical Science Collaboration at the RIKEN Brain Science Center to study aneurysms, while also working as chief of the Department of Neurosurgery at The University of Tokyo Hospital. In 2021, he left UTokyo Hospital to also become a professor at Kyorin University School of Medicine in Tokyo, also in conjunction with his work at RIKEN.
