Apr. 4, 2008 Research Highlight Biology
Splitting up cell fates during brain development
The orientation of the axis of progenitor cell division in the brain dictates whether daughter cells will maintain the parental cell fate
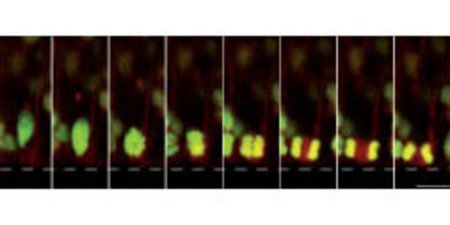 Figure 1: Neuroepithelial progenitor cell undergoing mitosis in the brain. Plane of division is parallel to the apicobasal axis and yet mitosis produces daughter cells with distinct cell fates. Green, chromosomes; red, cytoplasm. © Nature Cell Biology/ Nature Publishing Group/ 10/ 97 (2007)
Figure 1: Neuroepithelial progenitor cell undergoing mitosis in the brain. Plane of division is parallel to the apicobasal axis and yet mitosis produces daughter cells with distinct cell fates. Green, chromosomes; red, cytoplasm. © Nature Cell Biology/ Nature Publishing Group/ 10/ 97 (2007)
New findings indicate that the way in which the contents of progenitor cells called neuroepithelial cells are distributed during division strongly influences the fate of the resulting progeny. During division, the mitotic spindle is responsible for separating and allocating chromosomes into daughter cells. The spindle position dictates the orientation of the plane through which the parent cell will split.
Depending on the placement of the plane of division, progenitor cells can divide symmetrically—producing two daughter cells with fates identical to that of the parent cell, or asymmetrically—producing daughter cells having cell fates distinct from each other and/or from the parent cell. Daughter cells of neuroepithelial progenitor cells can either maintain the ‘parental’ progenitor fate, or embark along a developmental pathway leading to the neuron cell fate.
A team led by Fumio Matsuzaki at the RIKEN Center for Developmental Biology in Kobe conducted experiments to find out whether mitotic spindle orientation influenced the fate of neuroepithelial progenitors, as these cells are highly polarized with two distinct regions referred to as apical and basal endfoots. Their work was recently published in Nature Cell Biology 1.
By tracking the positions of fluorescently labeled chromosomes in neuroepithelial progenitors in developing mouse forebrains, the researchers noted that the majority of divisions were symmetric, or planar, along the plane perpendicular to the apicobasal axis (Fig. 1). To manipulate mitotic spindle orientation in neuroepithelial progenitors, the team ablated and enhanced expression of the proteins LGN and mInsc, respectively; both LGN and mInsc can shift spindle positioning.
Deletion of LGN and over-expression of mInsc resulted in asymmetric division occurring parallel to the apicobasal axis, and unequal distribution of apical and basal endfoots. Detailed study of individual dividing parent cells revealed that inheritance of both apical and basal endfoots was essential for adoption of the parental progenitor fate. Daughter cells inheriting either the apical or basal endfoots assumed the fate of cells on their way towards differentiating into neurons.
LGN ablation, which resulted in randomization of mitotic spindle positioning, led to depletion of neuroepithelial progenitors. Remarkably, however, overall neuron production remained unaltered.
Future study is needed to identify the cellular ‘cargo’ localized in the apical and basal endfoots of neuroepithelial cells that is required to bestow upon daughter cells a parental neuroepithelial progenitor cell destiny.
“There has been long-lasting controversy regarding mitotic spindle orientation during neuroepithelial progenitor divisions,” says Matsuzaki. “Our study provides a firm answer to this issue, and raises the fundamental question of how neurons are born from these planar divisions.”
References
- 1. Konno, D., Shioi, G., Shitamukai, A., Mori, A., Kiyonari, H., Miyata, T. & Matsuzaki, F. Neuroepithelial progenitors undergo LGN-dependent planar divisions to maintain self-renewability during mammalian neurogenesis. Nature Cell Biology 10, 93–101 (2007). doi: 10.1038/ncb1673
