Apr. 26, 2013 Research Highlight Biology
Managing the body’s interior architecture
Developing insects regulate growth of respiratory tubules through tightly controlled trafficking of an enzyme that remodels their interior infrastructure
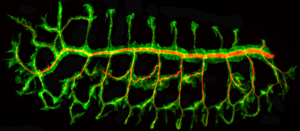 Figure 1: In the fly embryo, chitin polymer (red) accumulates within the luminal spaces of the developing tracheal network (green). © 2013 Shigeo Hayashi, RIKEN Center for Developmental Biology
Figure 1: In the fly embryo, chitin polymer (red) accumulates within the luminal spaces of the developing tracheal network (green). © 2013 Shigeo Hayashi, RIKEN Center for Developmental Biology
Virtually every multicellular organism requires the means to transport nutrients, water or other materials throughout the body. Tubule networks offer a simple mechanism for achieving this, although careful developmental regulation is required to ensure these tubules are of appropriate width and length to efficiently deliver their cargo. New research from a team led by Shigeo Hayashi of the Laboratory for Morphogenetic Signaling at the RIKEN Center for Developmental Biology now reveals an important mechanism governing the length of tubules in the tracheal network of flies1.
Fly respiration depends on proper formation of the system of tubules that comprise the tracheal network. Embryonic tracheal tubules are filled with a matrix of the polymer chitin, which accumulates within the interior, or lumen, of developing tubules (Fig. 1). This matrix is eventually cleared at the end of embryogenesis to make room for air to pass.
It is thought that a putative chitin-modifying enzyme called Serpentine (Serp) acts as a brake to limit tracheal tube elongation. During the growth process, Serp is taken up from the luminal space into the cells that form the tracheal tubules by a process called endocytosis. However, this is only temporary, and the internalized enzyme is eventually shuttled back to the lumen and reused. Hayashi’s team hypothesized that Rab-family proteins might facilitate this ‘recycling’ process. “These proteins are known to be crucial for membrane trafficking,” explains Hayashi, “and we searched for Rab proteins required for tracheal tube morphogenesis.”
They learned that embryos lacking Rab9 develop a trachea of excessive length. Examination of Serp localization in these embryos confirmed that this enzyme was being retained by tracheal cells after endocytosis, rather than being shuttled back into the luminal space. Intriguingly, tracheal tubule diameter was not altered in Rab9 mutant embryos, suggesting that diameter and length of tracheal tubules are regulated by separate mechanisms.
Subsequent experiments allowed Hayashi and his colleagues to identify numerous other proteins that collaborate with Rab9 in this process. Importantly, they were also able to observe key steps of the mechanism by which these proteins transfer endocytosed Serp into a ‘retrieval’ pathway that delivers it back to the lumen.
The team's findings could illuminate similar developmental mechanisms that occur in ‘higher’ organisms. “Chitin is present only in invertebrates and plants, but in a wider sense, vertebrate tubule organs like blood vessels and the neural tube are all formed through accumulation of luminal matrices,” says Hayashi. “It is an open field begging for intense investigation.”
References
- 1. Dong, B., Kakihara, K., Otani, T., Wada, H. & Hayashi, S. Rab9 and retromer regulate retrograde trafficking of luminal protein required for epithelial tube length control. Nature Communications 4, 1358 (2013). doi: 10.1038/ncomms2347
