Sep. 18, 2015 Research Highlight Biology
Controlling the cargo of cells
Insights into molecular transport and sorting systems in elongating cells may help prevent problems when things go wrong
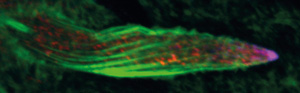 Figure 1: Spindle-F proteins (stained red) and IKKε proteins (stained blue) are localized toward the tip of Drosophila bristle cells. Modified from Ref. 1 and licensed under CC-BY 3.0 © 2015 T. Otani et al.
Figure 1: Spindle-F proteins (stained red) and IKKε proteins (stained blue) are localized toward the tip of Drosophila bristle cells. Modified from Ref. 1 and licensed under CC-BY 3.0 © 2015 T. Otani et al.
Through studying the cells of flies, RIKEN researchers have clarified the mechanisms by which cell components are transported in one direction during cell elongation1. These findings are expected to have wide relevance in biology and medicine.
‘Polarized’ cell elongation, in which differences between opposite ends or ‘poles’ of a cell are set up and maintained, is a crucial stage in the development of many cells and organisms. This stage requires the coordinated transport and selective retention of proteins and other molecules, but the molecular mechanisms underpinning these processes are poorly understood. Unraveling these processes is important because disruption of the transport system within cells can cause various diseases, including some involving the degeneration of nerve cells.
Now, Tetsuhisa Otani and his colleagues at the RIKEN Center for Developmental Biology, along with Uri Abdu at Ben-Gurion University in Israel, studied the elongation of the surface bristles of Drosophila flies to gain insights that have broad application. “Bristle cells are an excellent model system for understanding polarized cellular organization,” Otani explains, “and what we learn from this system might be extendable to other polarized cell types.”
A protein called IKKε regulates the organization of the bristle tips, but until now it was not clear how this protein became localized at the end of bristle cells and how it was kept there. Otani and his colleagues have found that a protein called Spindle-F grabs hold of the IKKε protein and anchors it on microtubules, which form a scaffolding within cells. Spindle-F is itself bound to the ‘motor protein’ dynein that moves cargo along the microtubules. The Spindle-F acts as an adaptor protein between dynein on the microtubules and the IKKε molecules. The researchers also found that a protein called Jvl regulates the retention of Spindle-F at the bristle tips.
“Our results suggest a basic regulatory principle for dynein-dependent transport,” says Otani, “and they have potential implications for understanding how disruption of this transport leads to neurodegenerative diseases.”
Identifying the molecules involved and their interactions is only one step toward understanding complex transport processes. An important challenge is to uncover how the Jvl protein regulates the system. Otani considers that the role of the molecular motor protein dynein may hold the key to revealing how cells do not suffer ‘lost luggage’—amazingly, all the required components somehow get transported to the correct places during the complex processes of cell development. He and his team intend to explore further the key aspects of how this is achieved.
References
- 1. Otani, T., Oshima, K., Kimpara, A., Takeda, M., Abdu, U. & Hayashi, S. A transport and retention mechanism for the sustained distal localization of Spn-F–IKKε during Drosophila bristle elongation. Development 142, 2338–2351 (2015). doi: 10.1242/dev.121863
