Dec. 16, 2016 Research Highlight Chemistry
A step toward unlocking the nitrogen reservoir in the sky
A titanium-based multi-metal complex turns inert nitrogen gas in the air into a reactive species that can be used to synthesize organic compounds
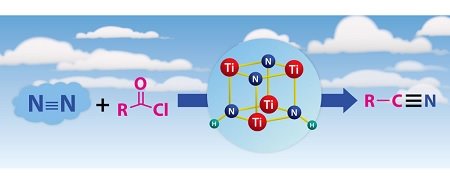 Figure 1: Atmospheric nitrogen (N2, blue) can be converted into useful organic nitriles (right) via the tetranuclear titanium nitrogen complex (compound in blue circle) in the presence of acyl chlorides (R(CO)Cl, pink). © 2016 Zhaomin Hou, RIKEN Center for Sustainable Resource Science
Figure 1: Atmospheric nitrogen (N2, blue) can be converted into useful organic nitriles (right) via the tetranuclear titanium nitrogen complex (compound in blue circle) in the presence of acyl chlorides (R(CO)Cl, pink). © 2016 Zhaomin Hou, RIKEN Center for Sustainable Resource Science
A complex containing four titanium atoms that can convert unreactive nitrogen gas (N2) into reactive species known as nitriles has been developed by RIKEN researchers. This may open the way to using atmospheric nitrogen for synthesizing useful nitrogen-containing organic compounds on industrial scales1.
Comprising about 78 per cent of the air we breathe, nitrogen is one of the most abundant chemicals on Earth. But the high stability of the nitrogen molecule makes it difficult to use atmospheric nitrogen as a feedstock for synthesizing organic chemicals. Since nitrogen is unreactive under normal conditions, the only industrial process that uses nitrogen gas as the starting material is energy intensive, requiring high temperatures (350 to 550 degrees Celsius) and pressures (150 to 350 atmospheres).
Zhaomin Hou from the RIKEN Center for Sustainable Resource Science and co-workers have succeeded in activating nitrogen under much milder conditions (60 degrees Celsius in benzene) by using a metal hydride complex that has four titanium centers.
“Multinuclear transition-metal hydrides, such as our tetranuclear complex, have multiple metal–hydrogen sites,” explains Hou. “They can thus provide more electrons and hydrogen atoms for cleaving and hydrogenating nitrogen than their mononuclear counterparts.”
Hou’s team generated the tetranuclear complex, which contains two types of nitrogen-containing units called imide (NH) and nitride (N), by reacting a titanium-based precursor with hydrogen gas (H2) in the presence of nitrogen (Fig. 1).
The researchers found that their complex split and partially hydrogenated nitrogen molecules without requiring extra reducing agents or proton sources. More importantly, the resulting nitrogen species reacted readily with acyl chlorides to produce organic nitriles under mild conditions without additional reagents, such as reducing agents or bases. “This is in sharp contrast with other approaches,” Hou comments.
Furthermore, the conversion of the nitrogen species into nitriles, which occurred in a single step, was compatible with functional groups that did not survive previous approaches. “We never anticipated that this transformation could occur in such a simple way,” says Hou. He notes that in earlier studies the conversion required using strong reducing agents and Lewis acids to remove oxygen and chlorine atoms by multi-step processes.
Having demonstrated that multinuclear titanium hydrides provide a promising platform for activating nitrogen and converting it into organic nitriles, the researchers are now exploring related compounds that have various metal–ligand combinations to better understand the relationship between molecular structure and reactivity. They plan to use this knowledge to develop nitrogen activation and functionalization systems with enhanced activity and efficiency.
Related contents
- Catalysis gets a metal-free upgrade
- Crystallizing the switch to hydrogen
- Green fixations for a cleaner future
References
- 1. Guru, M. M., Shima, T. & Hou, Z. Conversion of dinitrogen to nitriles at a multinuclear titanium framework. Angewandte Chemie International Edition 55, 12316–12320 (2016). doi: 10.1002/anie.201607426
