Dec. 14, 2022 Research Highlight Biology
An experimental strategy for recreating organ formation in the lab
Organ development has been modeled in the lab by selectively activating signaling pathways in the embryo
RIKEN researchers have imitated the formation of organs in the lab by ‘hacking’ certain signaling pathways that guide the development of connective tissues in the embryo, which support the formation of the liver, gut and other organs1. In addition to shedding light on organ development, this could eventually enable doctors to provide treatments tailored to individual patients.
The visceral organs are principally formed by embryonic epithelial cells—cells that line surfaces in the body. But recent research has revealed that this process involves close communication with the mesenchymal cells that ultimately give rise to the circulatory system, skeleton and various connective tissues (Fig. 1).
“Mesenchymal niche cells maintain epithelial stem cells, and the mesenchymal tissue structure influences the fates of epithelial cells,” explains Mitsuru Morimoto of the RIKEN Center for Biosystems Dynamics Research. This suggests that mesenchymal cells in various regions of the embryo provide distinct sets of instructions to the epithelial cells, which give rise to different organs.
In 2020, a team led by Aaron Zorn at Cincinnati Children’s Hospital systematically analyzed gene expression in the developing gut with single-cell resolution. They found specific signaling pathways that are active during early embryonic development, and which in turn give rise to mesenchymal lineages associated with different organs in the body.
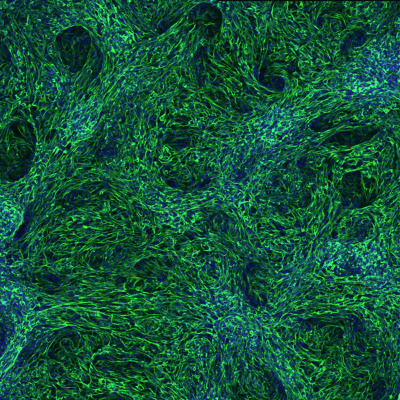
Figure 1: Liver-specific mesenchymal tissue derived from human stem cells. © 2022 RIKEN Center for Biosystems Dynamics Research
Now, by leveraging this knowledge, Morimoto and Zorn have developed a laboratory protocol that selectively activates these pathways to convert human stem cells into these various mesenchymal lineages. Using this approach, they were able to generate mesenchymal subtypes that help give rise to the liver, gastrointestinal tract or respiratory tissues after just one week of cell culture.
Keishi Kishimoto, a researcher from Morimoto’s group who recently joined Zorn’s lab, is enthusiastic about applying this approach in the context of regenerative medicine. “We can generate models of human congenital diseases in the lab using this protocol with patient-derived stem cells,” he says.
Cincinnati Children’s Hospital is a major hub for research into organoids—3D cell-culture models that closely mirror key structural and functional features of complex organs—and Kishimoto envisions using such systems to understand how various genetic mutations derail developmental processes.
To begin with, he and Morimoto will look at tracheoesophageal fistula, a rare condition in which the esophagus and windpipe are fused together, creating potentially serious respiratory issues. “We will generate tracheoesophageal fistula organoids in the laboratory by creating a foregut organoid and simultaneously inducing the trachea on one side and the esophagus on the other,” says Morimoto.
Related contents
- Reconstructing the cellular signaling pathways that shape trachea development
- Cultivating collectives of cells
Rate this article
Reference
- 1. Kishimoto, K., Iwasawa, K., Sorel, A., Ferran-Heredia, C., Han, L., Morimoto, M., Wells, J. M., Takebe, T. & Zorn, A. M. Directed differentiation of human pluripotent stem cells into diverse organ-specific mesenchyme of the digestive and respiratory systems. Nature Protocols 17, 2699–2719 (2022). doi: 10.1038/s41596-022-00733-3
